A physical linkage between cystic fibrosis airway surface dehydration and Pseudomonas aeruginosa biofilms
- PMID: 17116883
- PMCID: PMC1838718
- DOI: 10.1073/pnas.0606428103
A physical linkage between cystic fibrosis airway surface dehydration and Pseudomonas aeruginosa biofilms
Abstract
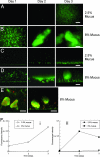
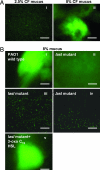
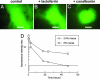
A vexing problem in cystic fibrosis (CF) pathogenesis has been to explain the high prevalence of Pseudomonas aeruginosa biofilms in CF airways. We speculated that airway surface liquid (ASL) hyperabsorption generates a concentrated airway mucus that interacts with P. aeruginosa to promote biofilms. To model CF vs. normal airway infections, normal (2.5% solids) and CF-like concentrated (8% solids) mucus were prepared, placed in flat chambers, and infected with an approximately 5 x 10(3) strain PAO1 P. aeruginosa. Although bacteria grew to 10(10) cfu/ml in both mucus concentrations, macrocolony formation was detected only in the CF-like (8% solids) mucus. Biophysical and functional measurements revealed that concentrated mucus exhibited properties that restrict bacterial motility and small molecule diffusion, resulting in high local bacterial densities with high autoinducer concentrations. These properties also rendered secondary forms of antimicrobial defense, e.g., lactoferrin, ineffective in preventing biofilm formation in a CF-like mucus environment. These data link airway surface liquid hyperabsorption to the high incidence of P. aeruginosa biofilms in CF via changes in the hydration-dependent physical-chemical properties of mucus and suggest that the thickened mucus gel model will be useful to develop therapies of P. aeruginosa biofilms in CF airways.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Matsui H, Grubb BR, Tarran R, Randell SH, Gatzy JT, Davis CW, Boucher RC. Cell. 1998;95:1005–1015. - PubMed
-
- Pedersen SS. APMIS Suppl. 1992;28:1–79. - PubMed
-
- Christensen BB, Sternberg C, Andersen JB, Palmer RJ, Nielsen AT, Givskov M, Molin S. In: Biofilms. Abelson JN, Simon MI, editors. Vol 310. San Diego: Academic; 1999. pp. 20–42.
-
- Davies DG, Parsek MR, Pearson JP, Iglewski BH, Costerton JW, Greenberg EP. Science. 1998;280:295–298. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

