TWEAK, via its receptor Fn14, is a novel regulator of mesenchymal progenitor cells and skeletal muscle regeneration
- PMID: 17124496
- PMCID: PMC1698888
- DOI: 10.1038/sj.emboj.7601441
TWEAK, via its receptor Fn14, is a novel regulator of mesenchymal progenitor cells and skeletal muscle regeneration
Abstract
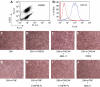
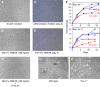
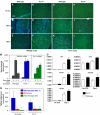
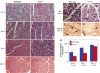
Inflammation participates in tissue repair through multiple mechanisms including directly regulating the cell fate of resident progenitor cells critical for successful regeneration. Upon surveying target cell types of the TNF ligand TWEAK, we observed that TWEAK binds to all progenitor cells of the mesenchymal lineage and induces NF-kappaB activation and the expression of pro-survival, pro-proliferative and homing receptor genes in the mesenchymal stem cells, suggesting that this pro-inflammatory cytokine may play an important role in controlling progenitor cell biology. We explored this potential using both the established C2C12 cell line and primary mouse muscle myoblasts, and demonstrated that TWEAK promoted their proliferation and inhibited their terminal differentiation. By generating mice deficient in the TWEAK receptor Fn14, we further showed that Fn14-deficient primary myoblasts displayed significantly reduced proliferative capacity and altered myotube formation. Following cardiotoxin injection, a known trigger for satellite cell-driven skeletal muscle regeneration, Fn14-deficient mice exhibited reduced inflammatory response and delayed muscle fiber regeneration compared with wild-type mice. These results indicate that the TWEAK/Fn14 pathway is a novel regulator of skeletal muscle precursor cells and illustrate an important mechanism by which inflammatory cytokines influence tissue regeneration and repair. Coupled with our recent demonstration that TWEAK potentiates liver progenitor cell proliferation, the expression of Fn14 on all mesenchymal lineage progenitor cells supports a broad involvement of this pathway in other tissue injury and disease settings.
Figures



References
-
- Arnett HA, Mason J, Marino M, Suzuki K, Matsushima GK, Ting JP (2001) TNF alpha promotes proliferation of oligodendrocyte progenitors and remyelination. Nat Neurosci 4: 1116–1122 - PubMed
-
- Barry FP (2003) Biology and clinical applications of mesenchymal stem cells. Birth Defects Res Part C Embryo Today 69: 250–256 - PubMed
-
- Bossen C, Ingold K, Tardivel A, Bodmer JL, Gaide O, Hertig S, Ambrose C, Tschopp J, Schneider P (2006) Interactions of tumor necrosis factor (TNF) and TNF receptor family members in the mouse and human. J Biol Chem 281: 13964–13971 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

