Putative role of beta-1,3 glucans in Candida albicans biofilm resistance
- PMID: 17130296
- PMCID: PMC1797745
- DOI: 10.1128/AAC.01056-06
Putative role of beta-1,3 glucans in Candida albicans biofilm resistance
Abstract
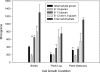
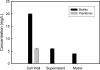
Biofilms are microbial communities, embedded in a polymeric matrix, growing attached to a surface. Nearly all device-associated infections involve growth in the biofilm life style. Biofilm communities have characteristic architecture and distinct phenotypic properties. The most clinically important phenotype involves extraordinary resistance to antimicrobial therapy, making biofilm infections very difficulty to cure without device removal. The current studies examine drug resistance in Candida albicans biofilms. Similar to previous reports, we observed marked fluconazole and amphotericin B resistance in a C. albicans biofilm both in vitro and in vivo. We identified biofilm-associated cell wall architectural changes and increased beta-1,3 glucan content in C. albicans cell walls from a biofilm compared to planktonic organisms. Elevated beta-1,3 glucan levels were also found in the surrounding biofilm milieu and as part of the matrix both from in vitro and in vivo biofilm models. We thus investigated the possible contribution of beta-glucans to antimicrobial resistance in Candida albicans biofilms. Initial studies examined the ability of cell wall and cell supernatant from biofilm and planktonic C. albicans to bind fluconazole. The cell walls from both environmental conditions bound fluconazole; however, four- to fivefold more compound was bound to the biofilm cell walls. Culture supernatant from the biofilm, but not planktonic cells, bound a measurable amount of this antifungal agent. We next investigated the effect of enzymatic modification of beta-1,3 glucans on biofilm cell viability and the susceptibility of biofilm cells to fluconazole and amphotericin B. We observed a dose-dependent killing of in vitro biofilm cells in the presence of three different beta-glucanase preparations. These same concentrations had no impact on planktonic cell viability. beta-1,3 Glucanase markedly enhanced the activity of both fluconazole and amphotericin B. These observations were corroborated with an in vivo biofilm model. Exogenous biofilm matrix and commercial beta-1,3 glucan reduced the activity of fluconazole against planktonic C. albicans in vitro. In sum, the current investigation identified glucan changes associated with C. albicans biofilm cells, demonstrated preferential binding of these biofilm cell components to antifungals, and showed a positive impact of the modification of biofilm beta-1,3 glucans on drug susceptibility. These results provide indirect evidence suggesting a role for glucans in biofilm resistance and present a strong rationale for further molecular dissection of this resistance mechanism to identify new drug targets to treat biofilm infections.
Figures


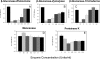

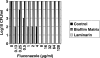
References
-
- Aketagawa, J., S. Tanaka, H. Tamura, Y. Shibata, and H. Saito. 1993. Activation of limulus coagulation factor G by several (1->3)-beta-D-glucans: comparison of the potency of glucans with identical degree of polymerization but different conformations. J. Biochem. 113:683-686. - PubMed
-
- Al-Fattani, M. A., and L. J. Douglas. 2006. Biofilm matrix of Candida albicans and Candida tropicalis: chemical composition and role in drug resistance. J. Med. Microbiol. 55:999-1008. - PubMed
-
- Amberg, D. C., D. J. Burke, J. N. Strathern, and D. Burke. 2005. Methods in yeast genetics: a Cold Spring Harbor Laboratory course manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.
-
- American Society for Microbiology. 2006. Antimicrobial Agents and Chemotherapy 2006 instructions to authors. Antimicrob. Agents Chemother. 50:1-21.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

