Perioperative strategy in colonic surgery; LAparoscopy and/or FAst track multimodal management versus standard care (LAFA trial)
- PMID: 17134506
- PMCID: PMC1693570
- DOI: 10.1186/1471-2482-6-16
Perioperative strategy in colonic surgery; LAparoscopy and/or FAst track multimodal management versus standard care (LAFA trial)
Abstract
Background: Recent developments in large bowel surgery are the introduction of laparoscopic surgery and the implementation of multimodal fast track recovery programs. Both focus on a faster recovery and shorter hospital stay. The randomized controlled multicenter LAFA-trial (LAparoscopy and/or FAst track multimodal management versus standard care) was conceived to determine whether laparoscopic surgery, fast track perioperative care or a combination of both is to be preferred over open surgery with standard care in patients having segmental colectomy for malignant disease.
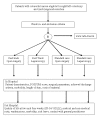
Methods/design: The LAFA-trial is a double blinded, multicenter trial with a 2 x 2 balanced factorial design. Patients eligible for segmental colectomy for malignant colorectal disease i.e. right and left colectomy and anterior resection will be randomized to either open or laparoscopic colectomy, and to either standard care or the fast track program. This factorial design produces four treatment groups; open colectomy with standard care (a), open colectomy with fast track program (b), laparoscopic colectomy with standard care (c), and laparoscopic surgery with fast track program (d). Primary outcome parameter is postoperative hospital length of stay including readmission within 30 days. Secondary outcome parameters are quality of life two and four weeks after surgery, overall hospital costs, morbidity, patient satisfaction and readmission rate. Based on a mean postoperative hospital stay of 9 +/- 2.5 days a group size of 400 patients (100 each arm) can reliably detect a minimum difference of 1 day between the four arms (alfa = 0.95, beta = 0.8). With 100 patients in each arm a difference of 10% in subscales of the Short Form 36 (SF-36) questionnaire and social functioning can be detected.
Discussion: The LAFA-trial is a randomized controlled multicenter trial that will provide evidence on the merits of fast track perioperative care and laparoscopic colorectal surgery in patients having segmental colectomy for malignant disease.
Figures
References
-
- Jacobs M, Verdeja JC, Goldstein HS. Minimally invasive colon resection (laparoscopic colectomy) Surg Laparosc Endosc. 1991;1:144–150. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical