Cytochrome C in a dry trehalose matrix: structural and dynamical effects probed by x-ray absorption spectroscopy
- PMID: 17142287
- PMCID: PMC1783899
- DOI: 10.1529/biophysj.106.092338
Cytochrome C in a dry trehalose matrix: structural and dynamical effects probed by x-ray absorption spectroscopy
Abstract
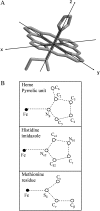
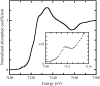
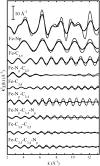
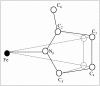
We report on the structure and dynamics of the Fe ligand cluster of reduced horse heart cytochrome c in solution, in a dried polyvinyl alcohol (PVA) film, and in two trehalose matrices characterized by different contents of residual water. The effect of the solvent/matrix environment was studied at room temperature using Fe K-edge x-ray absorption fine structure (XAFS) spectroscopy. XAFS data were analyzed by combining ab initio simulations and multi-parameter fitting in an attempt to disentangle structural from disorder parameters. Essentially the same structural and disorder parameters account adequately for the XAFS spectra measured in solution, both in the absence and in the presence of glycerol, and in the PVA film, showing that this polymer interacts weakly with the embedded protein. Instead, incorporation in trehalose leads to severe structural changes, more prominent in the more dried matrix, consisting of 1), an increase up to 0.2 A of the distance between Fe and the imidazole N atom of the coordinating histidine residue and 2), an elongation up to 0.16 A of the distance between Fe and the fourth-shell C atoms of the heme pyrrolic units. These structural distortions are accompanied by a substantial decrease of the relative mean-square displacements of the first ligands. In the extensively dried trehalose matrix, extremely low values of the Debye Waller factors are obtained for the pyrrolic and for the imidazole N atoms. This finding is interpreted as reflecting a drastic hindering in the relative motions of the Fe ligand cluster atoms and an impressive decrease in the static disorder of the local Fe structure. It appears, therefore, that the dried trehalose matrix dramatically perturbs the energy landscape of cytochrome c, giving rise, at the level of local structure, to well-resolved structural distortions and restricting the ensemble of accessible conformational substates.
Figures

Similar articles
-
The fe2+ site of photosynthetic reaction centers probed by multiple scattering x-ray absorption fine structure spectroscopy: improving structure resolution in dry matrices.Biophys J. 2008 Jul;95(2):814-22. doi: 10.1529/biophysj.108.132654. Epub 2008 May 2. Biophys J. 2008. PMID: 18456824 Free PMC article.
-
Electron transfer kinetics in photosynthetic reaction centers embedded in trehalose glasses: trapping of conformational substates at room temperature.Biophys J. 2002 Feb;82(2):558-68. doi: 10.1016/S0006-3495(02)75421-0. Biophys J. 2002. PMID: 11806901 Free PMC article.
-
Global mapping of structural solutions provided by the extended X-ray absorption fine structure ab initio code FEFF 6.01: structure of the cryogenic photoproduct of the myoglobin-carbon monoxide complex.Biochemistry. 1996 Jul 16;35(28):9014-23. doi: 10.1021/bi9605503. Biochemistry. 1996. PMID: 8703904
-
Structural heterogeneity of the Fe(2+)-N epsilon (HisF8) bond in various hemoglobin and myoglobin derivatives probed by the Raman-active iron histidine stretching mode.Biophys J. 1993 Oct;65(4):1470-85. doi: 10.1016/S0006-3495(93)81216-5. Biophys J. 1993. PMID: 8274641 Free PMC article.
-
Internal dynamics and protein-matrix coupling in trehalose-coated proteins.Biochim Biophys Acta. 2005 Jun 1;1749(2):252-81. doi: 10.1016/j.bbapap.2005.03.004. Epub 2005 Apr 15. Biochim Biophys Acta. 2005. PMID: 15886079 Review.
Cited by
-
X-Ray absorption studies of Zn2+ binding sites in bacterial, avian, and bovine cytochrome bc1 complexes.Biophys J. 2007 Oct 15;93(8):2934-51. doi: 10.1529/biophysj.107.110957. Epub 2007 Jun 15. Biophys J. 2007. PMID: 17573435 Free PMC article.
-
Proteins in amorphous saccharide matrices: structural and dynamical insights on bioprotection.Eur Phys J E Soft Matter. 2013 Jul;36(7):79. doi: 10.1140/epje/i2013-13079-x. Epub 2013 Jul 17. Eur Phys J E Soft Matter. 2013. PMID: 23884626
-
The fe2+ site of photosynthetic reaction centers probed by multiple scattering x-ray absorption fine structure spectroscopy: improving structure resolution in dry matrices.Biophys J. 2008 Jul;95(2):814-22. doi: 10.1529/biophysj.108.132654. Epub 2008 May 2. Biophys J. 2008. PMID: 18456824 Free PMC article.
References
-
- Uritani, M., M. Takai, and K. Yoshinaga. 1995. Protective effect of disaccharides on restriction endonucleases during drying under vacuum. J. Biochem. (Tokyo). 117:774–779. - PubMed
-
- Sun, W. Q., and P. Davidson. 1998. Protein inactivation in amorphous sucrose and trehalose matrices: effects of phase separation and crystallization. Biochim. Biophys. Acta. 1425:235–244. - PubMed
-
- Crowe, J. H., J. F. Carpenter, and L. M. Crowe. 1998. The role of vitrification in anhydrobiosis. Annu. Rev. Physiol. 60:73–103. - PubMed
-
- Crowe, L. M. 2002. Lessons from nature: the role of sugars in anhydrobiosis. Comp. Biochem. Physiol. A. 131:503–513. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

