Dopaminergic modulation of the persistence of one-trial hippocampus-dependent memory
- PMID: 17142305
- PMCID: PMC1783630
- DOI: 10.1101/lm.321006
Dopaminergic modulation of the persistence of one-trial hippocampus-dependent memory
Abstract
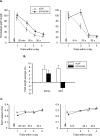
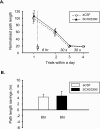
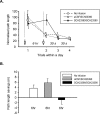
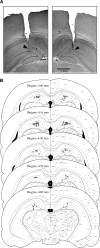
The persistence of new memory traces in the hippocampus, encoded following appropriate activation of glutamatergic receptors and the induction of synaptic plasticity, can be influenced by heterosynaptic activation of neuromodulatory brain systems. We therefore investigated the effects of a hippocampus-specific blockade of dopamine D1/D5 receptors on the persistence of spatial memory encoded in one trial using a delayed matching-to-place (DMP) task in a watermaze in which rats learn a new escape location each day. A within-subjects design was used such that both short (20 min) and long (6 h) retention intervals, and both drug (SCH23390, a D1/D5 receptor antagonist) and vehicle (aCSF) infusions were tested on different days in the same animals. Bilateral intrahippocampal infusion of SCH23390 (5 microg in 1 microL per side) prior to trial 1 (encoding) caused a differential impairment as a function of memory delay-with no effect during trial 2 (memory retrieval) after a 20-min interval, but a block of memory at 6 h. Further experiments revealed that infusion of SCH23390 immediately after trial 1 had no effect on retention 6 h later, and the poor memory seen at long retention intervals when the drug was present at encoding was not due to a state-dependent failure of retrieval. These results suggest that activation of D1/D5 receptors during memory encoding is necessary for the formation of a persistent memory trace in the hippocampus. The complementary effects of D1/D5 receptor blockade on the persistence of LTP and the duration of memory are consistent with the idea that changes in synaptic strength underlie memory.
Figures


Similar articles
-
Dopaminergic modulation of hippocampus-dependent learning: blockade of hippocampal D1-class receptors during learning impairs 1-trial place memory at a 30-min retention delay.Neuropharmacology. 2012 Sep;63(4):710-8. doi: 10.1016/j.neuropharm.2012.05.036. Epub 2012 May 30. Neuropharmacology. 2012. PMID: 22659087
-
D1/D5 dopamine receptors modulate spatial memory formation.Neurobiol Learn Mem. 2012 Feb;97(2):271-5. doi: 10.1016/j.nlm.2012.01.005. Epub 2012 Jan 14. Neurobiol Learn Mem. 2012. PMID: 22266268
-
Facilitation of fear extinction by novelty depends on dopamine acting on D1-subtype dopamine receptors in hippocampus.Proc Natl Acad Sci U S A. 2015 Mar 31;112(13):E1652-8. doi: 10.1073/pnas.1502295112. Epub 2015 Mar 9. Proc Natl Acad Sci U S A. 2015. PMID: 25775606 Free PMC article.
-
Dopamine D1/D5 receptors mediate informational saliency that promotes persistent hippocampal long-term plasticity.Cereb Cortex. 2014 Apr;24(4):845-58. doi: 10.1093/cercor/bhs362. Epub 2012 Nov 25. Cereb Cortex. 2014. PMID: 23183712 Free PMC article. Review.
-
Hippocampal synaptic plasticity: role in spatial learning or the automatic recording of attended experience?Philos Trans R Soc Lond B Biol Sci. 1997 Oct 29;352(1360):1489-503. doi: 10.1098/rstb.1997.0136. Philos Trans R Soc Lond B Biol Sci. 1997. PMID: 9368938 Free PMC article. Review.
Cited by
-
Post-training reward partially restores chronic stress induced effects in mice.PLoS One. 2012;7(6):e39033. doi: 10.1371/journal.pone.0039033. Epub 2012 Jun 22. PLoS One. 2012. PMID: 22745700 Free PMC article.
-
Navigating for reward.Nat Rev Neurosci. 2021 Aug;22(8):472-487. doi: 10.1038/s41583-021-00479-z. Epub 2021 Jul 6. Nat Rev Neurosci. 2021. PMID: 34230644 Free PMC article. Review.
-
Selectivity in Postencoding Connectivity with High-Level Visual Cortex Is Associated with Reward-Motivated Memory.J Neurosci. 2017 Jan 18;37(3):537-545. doi: 10.1523/JNEUROSCI.4032-15.2016. J Neurosci. 2017. PMID: 28100737 Free PMC article.
-
Role of G Protein-Coupled Receptors in the Regulation of Structural Plasticity and Cognitive Function.Molecules. 2017 Jul 24;22(7):1239. doi: 10.3390/molecules22071239. Molecules. 2017. PMID: 28737723 Free PMC article. Review.
-
Event boundaries drive norepinephrine release and distinctive neural representations of space in the rodent hippocampus.bioRxiv [Preprint]. 2024 Aug 31:2024.07.30.605900. doi: 10.1101/2024.07.30.605900. bioRxiv. 2024. PMID: 39131365 Free PMC article. Preprint.
References
-
- Abe K. Modulation of hippocampal long-term potentiation by the amygdala: A synaptic mechanism linking emotion and memory. Jpn. J. Pharmacol. 2001;86:18–22. - PubMed
-
- Adcock R.A., Thangavel A., Whitfield-Gabrieli S., Knutson B., Gabrieli J.D.E. Reward-motivated learning: Mesolimbic activation precedes memory formation. Neuron. 2006;50:507–517. - PubMed
-
- Aggleton J.P., Brown M.W. Episodic memory, amnesia, and the hippocampal-anterior thalamic axis. Behav. Brain Sci. 1999;22:425–444. - PubMed
-
- Almaguer-Melian W., Martinez-Marti L., Frey J.U., Bergado J.A. The amygdala is part of the behavioural reinforcement system modulating long-term potentiation in rat hippocampus. Neuroscience. 2003;119:319–322. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
