X-ray crystallographic study of DNA duplex cross-linking: simultaneous binding to two d(CGTACG)2 molecules by a bis(9-aminoacridine-4-carboxamide) derivative
- PMID: 17145714
- PMCID: PMC1751537
- DOI: 10.1093/nar/gkl930
X-ray crystallographic study of DNA duplex cross-linking: simultaneous binding to two d(CGTACG)2 molecules by a bis(9-aminoacridine-4-carboxamide) derivative
Abstract
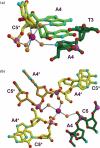
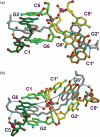
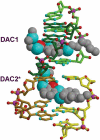
Acridine-4-carboxamides form a class of known DNA mono-intercalating agents that exhibit cytotoxic activity against tumour cell lines due to their ability to inhibit topoisomerases. Previous studies of bis-acridine derivatives have yielded equivocal results regarding the minimum length of linker necessary between the two acridine chromophores to allow bis-intercalation of duplex DNA. We report here the 1.7 A resolution X-ray crystal structure of a six-carbon-linked bis(acridine-4-carboxamide) ligand bound to d(CGTACG)2 molecules by non-covalent duplex cross-linking. The asymmetric unit consists of one DNA duplex containing an intercalated acridine-4-carboxamide chromophore at each of the two CG steps. The other half of each ligand is bound to another DNA molecule in a symmetry-related manner, with the alkyl linker threading through the minor grooves. The two crystallographically independent ligand molecules adopt distinct side chain interactions, forming hydrogen bonds to either O6 or N7 on the major groove face of guanine, in contrast to the semi-disordered state of mono-intercalators bound to the same DNA molecule. The complex described here provides the first structural evidence for the non-covalent cross-linking of DNA by a small molecule ligand and suggests a possible explanation for the inconsistent behaviour of six-carbon linked bis-acridines in previous assays of DNA bis-intercalation.
Figures



Similar articles
-
Crystal structure of the topoisomerase II poison 9-amino-[N-(2-dimethylamino)ethyl]acridine-4-carboxamide bound to the DNA hexanucleotide d(CGTACG)2.Biochemistry. 1999 Jul 20;38(29):9221-33. doi: 10.1021/bi990352m. Biochemistry. 1999. PMID: 10413496
-
Structure of 9-amino-[N-(2-dimethylamino)propyl]acridine-4-carboxamide bound to d(CGTACG)(2): a comparison of structures of d(CGTACG)(2) complexed with intercalatorsin the presence of cobalt.Acta Crystallogr D Biol Crystallogr. 2004 May;60(Pt 5):823-8. doi: 10.1107/S0907444904003907. Epub 2004 Apr 21. Acta Crystallogr D Biol Crystallogr. 2004. PMID: 15103127
-
Structural characterisation of bisintercalation in higher-order DNA at a junction-like quadruplex.J Mol Biol. 2002 Oct 18;323(2):167-71. doi: 10.1016/s0022-2836(02)00923-3. J Mol Biol. 2002. PMID: 12381312
-
Genotoxicity of non-covalent interactions: DNA intercalators.Mutat Res. 2007 Oct 1;623(1-2):14-23. doi: 10.1016/j.mrfmmm.2007.03.014. Epub 2007 Apr 6. Mutat Res. 2007. PMID: 17498749 Review.
-
Crystal structures of acridines complexed with nucleic acids.Curr Med Chem. 2002 Sep;9(18):1667-75. doi: 10.2174/0929867023369259. Curr Med Chem. 2002. PMID: 12171549 Review.
Cited by
-
Interactions of small molecules with DNA junctions.Nucleic Acids Res. 2022 Dec 9;50(22):12636-12656. doi: 10.1093/nar/gkac1043. Nucleic Acids Res. 2022. PMID: 36382400 Free PMC article. Review.
-
Zinc Complexes with Nitrogen Donor Ligands as Anticancer Agents.Molecules. 2020 Dec 9;25(24):5814. doi: 10.3390/molecules25245814. Molecules. 2020. PMID: 33317158 Free PMC article. Review.
-
Studies on 1,4-Quinone Derivatives Exhibiting Anti-Leukemic Activity along with Anti-Colorectal and Anti-Breast Cancer Effects.Molecules. 2022 Dec 22;28(1):77. doi: 10.3390/molecules28010077. Molecules. 2022. PMID: 36615273 Free PMC article.
-
Supramolecular Recognition of a DNA Four-Way Junction by an M2L4 Metallo-Cage, Inspired by a Simulation-Guided Design Approach.Angew Chem Int Ed Engl. 2025 Jun 24;64(26):e202504866. doi: 10.1002/anie.202504866. Epub 2025 May 5. Angew Chem Int Ed Engl. 2025. PMID: 40243103 Free PMC article.
-
C 3-symmetric opioid scaffolds are pH-responsive DNA condensation agents.Nucleic Acids Res. 2017 Jan 25;45(2):527-540. doi: 10.1093/nar/gkw1097. Epub 2016 Nov 28. Nucleic Acids Res. 2017. PMID: 27899572 Free PMC article.
References
-
- Waring M.J., Wakelin L.P. Echinomycin: a bifunctional intercalating antibiotic. Nature. 1974;252:653–657. - PubMed
-
- Wang A.H., Ughetto G., Quigley G.J., Hakoshima T., van der Marel G.A., van Boom J.H., Rich A. The molecular structure of a DNA–triostin A complex. Science. 1984;225:1115–1121. - PubMed
-
- Wakelin L.P.G., Romanos M., Chen T.K., Glaubiger D., Canellakis E.S., Waring M.J. Structural limitations on the bifunctional intercalation of diacridines into DNA. Biochemistry. 1978;17:5057–5063. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

