APOBEC3G/3F mediates intrinsic resistance of monocyte-derived dendritic cells to HIV-1 infection
- PMID: 17145955
- PMCID: PMC2118170
- DOI: 10.1084/jem.20061519
APOBEC3G/3F mediates intrinsic resistance of monocyte-derived dendritic cells to HIV-1 infection
Abstract
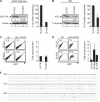
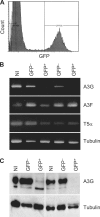
HIV-1 infects immature dendritic cells (iDCs), but infection is inefficient compared with activated CD4+ T cells and only involves a small subset of iDCs. We analyzed whether this could be attributed to specific cellular restrictions during the viral life cycle. To study env-independent restriction to HIV-1 infection, we used a single-round infection assay with HIV-1 pseudotyped with vesicular stomatitis virus G protein (HIV-VSVG). Small interfering RNA-mediated depletion of APOBEC3G/3F (A3G/3F), but not TRIM5alpha, enhanced HIV-1 infection of iDCs, indicating that A3G/3F controls the sensitivity of iDCs to HIV-1 infection. Furthermore, sequences of HIV reverse transcripts revealed G-to-A hypermutation of HIV genomes during iDC infection, demonstrating A3G/3F cytidine deaminase activity in iDCs. When we separated the fraction of iDCs that was susceptible to HIV, we found the cells to be deficient in A3G messenger RNA and protein. We also noted that during DC maturation, which further reduces susceptibility to infection, A3G levels increased. These findings highlight a role for A3G/3F in explaining the resistance of most DCs to HIV-1 infection, as well as the susceptibility of a fraction of iDCs. An increase in the A3G/3F-mediated intrinsic resistance of iDCs could result in a block of HIV infection at its mucosal point of entry.
Figures
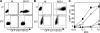


References
-
- Piguet, V., and A. Blauvelt. 2002. Essential roles for dendritic cells in the pathogenesis and potential treatment of HIV disease. J. Invest. Dermatol. 119:365–369. - PubMed
-
- Steinman, R.M., A. Granelli-Piperno, M. Pope, C. Trumpfheller, R. Ignatius, G. Arrode, P. Racz, and K. Tenner-Racz. 2003. The interaction of immunodeficiency viruses with dendritic cells. Curr. Top. Microbiol. Immunol. 276:1–30. - PubMed
-
- Margolis, L., and R. Shattock. 2006. Selective transmission of CCR5-utilizing HIV-1: the ‘gatekeeper’ problem resolved? Nat. Rev. Microbiol. 4:312–317. - PubMed
-
- Granelli-Piperno, A., E. Delgado, V. Finkel, W. Paxton, and R.M. Steinman. 1998. Immature dendritic cells selectively replicate macrophagetropic (M-tropic) human immunodeficiency virus type 1, while mature cells efficiently transmit both M- and T-tropic virus to T cells. J. Virol. 72:2733–2737. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

