Tumor necrosis factor-alpha mediates oligodendrocyte death and delayed retinal ganglion cell loss in a mouse model of glaucoma
- PMID: 17151265
- PMCID: PMC6674838
- DOI: 10.1523/JNEUROSCI.2801-06.2006
Tumor necrosis factor-alpha mediates oligodendrocyte death and delayed retinal ganglion cell loss in a mouse model of glaucoma
Abstract
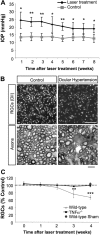
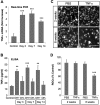
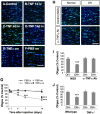
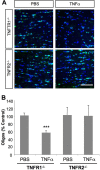
Glaucoma is a widespread ocular disease characterized by a progressive loss of retinal ganglion cells (RGCs). Previous studies suggest that the cytokine tumor necrosis factor-alpha (TNF-alpha) may contribute to the disease process, although its role in vivo and its mechanism of action are unclear. To investigate pathophysiological mechanisms in glaucoma, we induced ocular hypertension (OH) in mice by angle closure via laser irradiation. This treatment resulted in a rapid upregulation of TNF-alpha, followed sequentially by microglial activation, loss of optic nerve oligodendrocytes, and delayed loss of RGCs. Intravitreal TNF-alpha injections in normal mice mimicked these effects. Conversely, an anti-TNF-alpha-neutralizing antibody or deleting the genes encoding TNF-alpha or its receptor, TNFR2, blocked the deleterious effects of OH. Deleting the CD11b/CD18 gene prevented microglial activation and also blocked the pathophysiological effects of OH. Thus TNF-alpha provides an essential, although indirect, link between OH and RGC loss in vivo. Blocking TNF-alpha signaling or inflammation, therefore, may be helpful in treating glaucoma.
Figures




References
-
- Ahmed F, Brown KM, Stephan DA, Morrison JC, Johnson EC, Tomarev SI. Microarray analysis of changes in mRNA levels in the rat retina after experimental elevation of intraocular pressure. Invest Ophthalmol Vis Sci. 2004;45:1247–1258. - PubMed
-
- Aihara M, Lindsey JD, Weinreb RN. Experimental mouse ocular hypertension: establishment of the model. Invest Ophthalmol Vis Sci. 2003;44:4314–4320. - PubMed
-
- Alldred A. Etanercept in rheumatoid arthritis. Expert Opin Pharmacother. 2001;2:1137–1148. - PubMed
-
- Bohatschek M, Kloss CU, Hristova M, Pfeffer K, Raivich G. Microglial major histocompatibility complex glycoprotein-1 in the axotomized facial motor nucleus: regulation and role of tumor necrosis factor receptors 1 and 2. J Comp Neurol. 2004;470:382–399. - PubMed
-
- Brouckaert P, Libert C, Everaerdt B, Takahashi N, Cauwels A, Fiers W. Tumor necrosis factor, its receptors and the connection with interleukin 1 and interleukin 6. Immunobiology. 1993;187:317–329. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
