Advances in combating fungal diseases: vaccines on the threshold
- PMID: 17160002
- PMCID: PMC2214303
- DOI: 10.1038/nrmicro1537
Advances in combating fungal diseases: vaccines on the threshold
Abstract
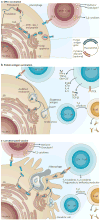
The dramatic increase in fungal diseases in recent years can be attributed to the increased aggressiveness of medical therapy and other human activities. Immunosuppressed patients are at risk of contracting fungal diseases in healthcare settings and from natural environments. Increased prescribing of antifungals has led to the emergence of resistant fungi, resulting in treatment challenges. These concerns, together with the elucidation of the mechanisms of protective immunity against fungal diseases, have renewed interest in the development of vaccines against the mycoses. Most research has used murine models of human disease and, as we review in this article, the knowledge gained from these studies has advanced to the point where the development of vaccines targeting human fungal pathogens is now a realistic and achievable goal.
Figures


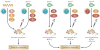

References
-
- Mochon AB, Cutler JE. Is a vaccine needed against Candida albicans? Med Mycol. 2005;43:97–115. - PubMed
-
- Deepe GS, Jr, Wüthrich M, Klein BS. Progress in vaccination for histoplasmosis and blastomycosis: coping with cellular immunity. Med Mycol. 2005;43:381–389. - PubMed
-
- Cole GT, et al. A vaccine against coccidioidomycosis is justified and attainable. Med Mycol. 2004;42:189–216. This work is an outstanding contemporary review of the topic. - PubMed
-
- Deepe GS., Jr Preventative and therapeutic vaccines for fungal infections: from concept to implementation. Expert Rev Vaccines. 2004;3:1–9. - PubMed
-
- Casadevall A, Pirofski LA. Feasibility and prospects for a vaccine to prevent cryptococcosis. Med Mycol. 2005;43:667–680. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

