For a healthy histone code, a little SUMO in the tail keeps the acetyl away
- PMID: 17163672
- PMCID: PMC2170887
- DOI: 10.1021/cb600188m
For a healthy histone code, a little SUMO in the tail keeps the acetyl away
Abstract
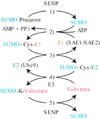
Chemical modification of histones through a growing number of post-translational mechanisms is an integral part of transcription. A recent report provides exciting new evidence that conjugation of the ubiquitin-like protein SUMO to histones opposes acetylation and establishes SUMOylation as an important histone mark linked to transcriptional repression.
Figures

Similar articles
-
Identification of a new small ubiquitin-like modifier (SUMO)-interacting motif in the E3 ligase PIASy.J Biol Chem. 2017 Jun 16;292(24):10230-10238. doi: 10.1074/jbc.M117.789982. Epub 2017 Apr 28. J Biol Chem. 2017. PMID: 28455449 Free PMC article.
-
SUMO association with repressor complexes, emerging routes for transcriptional control.Biochim Biophys Acta. 2009 Jun-Aug;1789(6-8):451-9. doi: 10.1016/j.bbagrm.2009.07.001. Epub 2009 Jul 17. Biochim Biophys Acta. 2009. PMID: 19616654 Review.
-
SUMO and transcriptional regulation.Semin Cell Dev Biol. 2004 Apr;15(2):201-10. doi: 10.1016/j.semcdb.2003.12.001. Semin Cell Dev Biol. 2004. PMID: 15209380 Review.
-
Molecular mechanisms in SUMO conjugation.Biochem Soc Trans. 2020 Feb 28;48(1):123-135. doi: 10.1042/BST20190357. Biochem Soc Trans. 2020. PMID: 31872228 Review.
-
Analysis of Histone Deacetylases Sumoylation by Immunoprecipitation Techniques.Methods Mol Biol. 2017;1510:339-351. doi: 10.1007/978-1-4939-6527-4_25. Methods Mol Biol. 2017. PMID: 27761833
Cited by
-
A comprehensive view of the epigenetic landscape. Part II: Histone post-translational modification, nucleosome level, and chromatin regulation by ncRNAs.Neurotox Res. 2015 Feb;27(2):172-97. doi: 10.1007/s12640-014-9508-6. Epub 2014 Dec 17. Neurotox Res. 2015. PMID: 25516120 Free PMC article. Review.
-
Epigenetic mechanisms facilitating oligodendrocyte development, maturation, and aging.Glia. 2009 Nov 15;57(15):1579-87. doi: 10.1002/glia.20881. Glia. 2009. PMID: 19373939 Free PMC article. Review.
-
Viral Mimicry to Usurp Ubiquitin and SUMO Host Pathways.Viruses. 2015 Aug 28;7(9):4854-72. doi: 10.3390/v7092849. Viruses. 2015. PMID: 26343706 Free PMC article. Review.
-
Regulation of cardiac specific nkx2.5 gene activity by small ubiquitin-like modifier.J Biol Chem. 2008 Aug 22;283(34):23235-43. doi: 10.1074/jbc.M709748200. Epub 2008 Jun 24. J Biol Chem. 2008. PMID: 18579533 Free PMC article.
-
An epigenetic timer regulates the transition from cell division to cell expansion during Arabidopsis petal organogenesis.PLoS Genet. 2024 Mar 5;20(3):e1011203. doi: 10.1371/journal.pgen.1011203. eCollection 2024 Mar. PLoS Genet. 2024. PMID: 38442104 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

