In vitro polymerization of a functional Escherichia coli amyloid protein
- PMID: 17164238
- PMCID: PMC2838475
- DOI: 10.1074/jbc.M609228200
In vitro polymerization of a functional Escherichia coli amyloid protein
Abstract
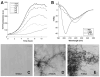
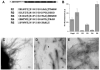
Amyloid formation is characterized by the conversion of soluble proteins into biochemically and structurally distinct fibers. Although amyloid formation is traditionally associated with diseases such as Alzheimer disease, a number of biologically functional amyloids have recently been described. Curli are amyloid fibers produced by Escherichia coli that contribute to biofilm formation and other important physiological processes. We characterized the polymerization properties of the major curli subunit protein CsgA. CsgA polymerizes into an amyloid fiber in a sigmoidal kinetic fashion with a distinct lag, growth, and stationary phase. Adding sonicated preformed CsgA fibers to the polymerization reaction can significantly shorten the duration of the lag phase. We also demonstrate that the conversion of soluble CsgA into an insoluble fiber involves the transient formation of an intermediate similar to that characterized for several disease-associated amyloids. The CsgA core amyloid domain can be divided into five repeating units that share sequence and structural hallmarks. We show that peptides representing three of these repeating units are amyloidogenic in vitro. Although the defining characteristics of CsgA polymerization appear conserved with disease-associated amyloids, these proteins evolved in diverse systems and for different purposes. Therefore, amyloidogenesis appears to be an innate protein folding pathway that can be capitalized on to fulfill normal physiological tasks.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

