Cell-free production of scFv fusion proteins: an efficient approach for personalized lymphoma vaccines
- PMID: 17164345
- PMCID: PMC1852255
- DOI: 10.1182/blood-2006-07-030593
Cell-free production of scFv fusion proteins: an efficient approach for personalized lymphoma vaccines
Abstract
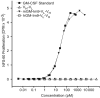
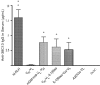
The unique immunoglobulin (Ig) idiotype on the surface of each B-cell lymphoma represents an ideal tumor-specific antigen for use as a therapeutic vaccine. We have used an Escherichia coli-based, cell-free protein-expression system to produce a vaccine within hours of cloning the Ig genes from a B-cell tumor. We demonstrated that a fusion protein consisting of an idiotypic single chain Fv antibody fragment (scFv) linked to a cytokine (GM-CSF) or to an immunostimulatory peptide was an effective lymphoma vaccine. These vaccines elicited humoral immune responses against the native Ig protein displayed on the surface of a tumor and protected mice against tumor challenge with efficacy equal to that of the conventional Ig produced in a mammalian cell and chemically coupled to keyhole limpet hemocyanin. The cell-free E coli system offers a platform for rapidly generating individualized vaccines, thereby allowing much more efficient application in the clinic.
Figures
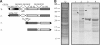


Similar articles
-
A genetic approach to idiotypic vaccination for B cell lymphoma.Ann N Y Acad Sci. 1995 Nov 27;772:212-26. doi: 10.1111/j.1749-6632.1995.tb44747.x. Ann N Y Acad Sci. 1995. PMID: 8546396 Review.
-
Immunization with a recombinant adenovirus encoding a lymphoma idiotype: induction of tumor-protective immunity and identification of an idiotype-specific T cell epitope.J Immunol. 2002 Apr 15;168(8):3983-91. doi: 10.4049/jimmunol.168.8.3983. J Immunol. 2002. PMID: 11937555
-
[Construction and expression of a prokaryotic expression plasmid of idiotypic vaccine against B cell lymphoma: encoding the fusion genes of single-chain variable fragment and MCP-3].Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2006 Dec;14(6):1151-5. Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2006. PMID: 17204183 Chinese.
-
Escherichia coli-based production of a tumor idiotype antibody fragment--tetanus toxin fragment C fusion protein vaccine for B cell lymphoma.Protein Expr Purif. 2011 Jan;75(1):15-20. doi: 10.1016/j.pep.2010.09.005. Epub 2010 Sep 19. Protein Expr Purif. 2011. PMID: 20851769
-
Therapeutic lymphoma vaccines: importance of T-cell immunity.Expert Rev Vaccines. 2006 Jun;5(3):381-94. doi: 10.1586/14760584.5.3.381. Expert Rev Vaccines. 2006. PMID: 16827622 Review.
Cited by
-
Deconstructing Cell-Free Extract Preparation for in Vitro Activation of Transcriptional Genetic Circuitry.ACS Synth Biol. 2019 Feb 15;8(2):403-414. doi: 10.1021/acssynbio.8b00430. Epub 2019 Jan 29. ACS Synth Biol. 2019. PMID: 30596483 Free PMC article.
-
Cell-Free Approaches in Synthetic Biology Utilizing Microfluidics.Genes (Basel). 2018 Mar 6;9(3):144. doi: 10.3390/genes9030144. Genes (Basel). 2018. PMID: 29509709 Free PMC article. Review.
-
Applications of synthetic biology in medical and pharmaceutical fields.Signal Transduct Target Ther. 2023 May 11;8(1):199. doi: 10.1038/s41392-023-01440-5. Signal Transduct Target Ther. 2023. PMID: 37169742 Free PMC article. Review.
-
Screening for a single-chain variable-fragment antibody that can effectively neutralize the cytotoxicity of the Vibrio parahaemolyticus thermolabile hemolysin.Appl Environ Microbiol. 2012 Jul;78(14):4967-75. doi: 10.1128/AEM.00435-12. Epub 2012 May 4. Appl Environ Microbiol. 2012. PMID: 22562997 Free PMC article.
-
Cell-free production of Gaussia princeps luciferase--antibody fragment bioconjugates for ex vivo detection of tumor cells.Biochem Biophys Res Commun. 2009 Dec 18;390(3):971-6. doi: 10.1016/j.bbrc.2009.10.087. Epub 2009 Oct 21. Biochem Biophys Res Commun. 2009. PMID: 19852937 Free PMC article.
References
-
- Brown SL, Miller RA, Horning SJ, et al. Treatment of B-cell lymphomas with anti-idiotype antibodies alone and in combination with alpha interferon. Blood. 1989;73:651–661. - PubMed
-
- Levy R, Miller RA. Tumor therapy with monoclonal antibodies. Fed Proc. 1983;42:2650–2656. - PubMed
-
- Maloney DG, Brown S, Czerwinski DK, et al. Monoclonal anti-idiotype antibody therapy of B-cell lymphoma: the addition of a short course of chemotherapy does not interfere with the antitumor effect nor prevent the emergence of idiotype-negative variant cells. Blood. 1992;80:1502–1510. - PubMed
-
- Meeker TC, Lowder J, Maloney DG, et al. A clinical trial of anti-idiotype therapy for B cell malignancy. Blood. 1985;65:1349–1363. - PubMed
-
- Miller RA, Maloney DG, Warnke R, Levy R. Treatment of B-cell lymphoma with monoclonal anti-idiotype antibody. N Engl J Med. 1982;306:517–522. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

