De novo formal synthesis of (-)-apicularen A via an iterative asymmetric hydration sequence
- PMID: 17165936
- PMCID: PMC2529256
- DOI: 10.1021/ol062595u
De novo formal synthesis of (-)-apicularen A via an iterative asymmetric hydration sequence
Abstract
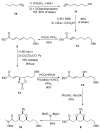
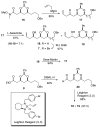
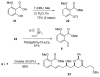
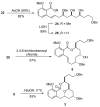
[Structure: see text] A de novo approach to the formal total synthesis of the macrolide natural product (-)-apicularen A has been achieved in 18 steps from achiral starting materials. Both the absolute and relative stereochemistries of apicularen A were introduced by a Sharpless asymmetric dihydroxylation, a pi-allyl-palladium catalyzed reduction, a stereoselective reduction, and a base-promoted transannulation to install the C-9 stereocenter.
Figures


Similar articles
-
De novo synthesis of natural products via the asymmetric hydration of polyenes.Chem Commun (Camb). 2011 Aug 14;47(30):8493-505. doi: 10.1039/c1cc11791b. Epub 2011 May 11. Chem Commun (Camb). 2011. PMID: 21559534 Free PMC article. Review.
-
De Novo Asymmetric Synthesis of (+)-Monanchorin.Org Lett. 2015 Nov 6;17(21):5280-3. doi: 10.1021/acs.orglett.5b02651. Epub 2015 Oct 13. Org Lett. 2015. PMID: 26461800
-
Total synthesis and biological evaluation of (-)-apicularen A and its analogues.J Org Chem. 2012 Jan 6;77(1):388-99. doi: 10.1021/jo2019762. Epub 2011 Dec 6. J Org Chem. 2012. PMID: 22118352
-
Establishment of relative and absolute configurations of phaeosphaeride A: total synthesis of ent-phaeosphaeride A.J Org Chem. 2015 Jan 16;80(2):1243-8. doi: 10.1021/jo5025046. Epub 2015 Jan 7. J Org Chem. 2015. PMID: 25517514
-
De novo asymmetric synthesis of the pyranoses-from monosaccharides to oligosaccharides: An update.Adv Carbohydr Chem Biochem. 2024;85:1-38. doi: 10.1016/bs.accb.2024.10.004. Epub 2024 Nov 8. Adv Carbohydr Chem Biochem. 2024. PMID: 39572133 Review.
Cited by
-
Total synthesis of fostriecin: via a regio- and stereoselective polyene hydration, oxidation, and hydroboration sequence.Org Lett. 2010 Sep 3;12(17):3752-5. doi: 10.1021/ol101340n. Org Lett. 2010. PMID: 20687585 Free PMC article.
-
De Novo Asymmetric Syntheses of (+)-Goniothalamin, (+)-Goniothalamin oxide and 7,8-Bis-epi-Goniothalamin using Asymmetric Allylations.Tetrahedron. 2009 Jun 27;65(26):5051-5055. doi: 10.1016/j.tet.2009.03.097. Tetrahedron. 2009. PMID: 20161297 Free PMC article.
-
Cryptocaryols A and B: total syntheses, stereochemical revision, structure elucidation, and structure-activity relationship.J Am Chem Soc. 2013 Jun 26;135(25):9334-7. doi: 10.1021/ja404401f. Epub 2013 Jun 14. J Am Chem Soc. 2013. PMID: 23750754 Free PMC article.
-
De novo synthesis of natural products via the asymmetric hydration of polyenes.Chem Commun (Camb). 2011 Aug 14;47(30):8493-505. doi: 10.1039/c1cc11791b. Epub 2011 May 11. Chem Commun (Camb). 2011. PMID: 21559534 Free PMC article. Review.
-
Identification of α2 macroglobulin as a major serum ghrelin esterase.Angew Chem Int Ed Engl. 2011 Nov 4;50(45):10699-702. doi: 10.1002/anie.201104512. Epub 2011 Sep 16. Angew Chem Int Ed Engl. 2011. PMID: 21928452 Free PMC article. No abstract available.
References
-
- Kunze B, Jansen R, Sasse F, Höfle G, Reichenbach H. J. Antibiot. 1998;51:1075–1080. - PubMed
- Jansen R, Kunze B, Reichenbach H, Höfle G. Eur. J. Org. Chem. 2000:913–919.
-
- Bhattacharjee A, Seguil OR, De Brabander JK. Tetrahedron Lett. 2001;42:1217–1220.
- Nicolaou KC, Kim DW, Baati R. Angew. Chem. Int. Ed. 2002;41:3701–3704. - PubMed
- Su Q, Panek JS. J. Am. Chem. Soc. 2004;126:2425–2430. - PubMed
- Petri AF, Bayer A, Maier ME. Angew. Chem. Int. Ed. 2004;43:5821–5823. - PubMed
-
- Lewis A, Stefanuti I, Swain SA, Smith SA, Taylor RJK. Tetrahedron Lett. 2001;42:5549–5552.
- Lewis A, Stefanuti I, Swain SA, Smith SA, Taylor RJK. Org. Biomol. Chem. 2003;1:104–116. - PubMed
- Graetz BR, Rychnovsky SD. Org. Lett. 2003;5:3357–3360. - PubMed
- Kühnert S, Maier ME. Org. Lett. 2002;4:643–646. - PubMed
- Hilli F, White JM, Rizzacasa MA. Tetrahedron Lett. 2002;43:8507–8510.
- Hilli F, White JM, Rizzacasa MA. Org. Lett. 2004;6:1289–1292. - PubMed
-
-
While Maier’s endgame seemed ideal for our purpose, his use of a stochiometric amount (CF3CO2)2Hg to set the trans-annular ether bridge in macrolide 3 (see ref 3d) was viewed as needing to be replaced with an environmentally more benign yet equally stereoselective process.
-
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

