Dosage-dependent switch from G protein-coupled to G protein-independent signaling by a GPCR
- PMID: 17170700
- PMCID: PMC1782364
- DOI: 10.1038/sj.emboj.7601502
Dosage-dependent switch from G protein-coupled to G protein-independent signaling by a GPCR
Abstract
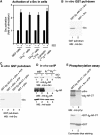
G-protein-coupled receptors (GPCRs) mostly signal through heterotrimeric G proteins. Increasing evidence suggests that GPCRs could function in a G-protein-independent manner. Here, we show that at low concentrations of an agonist, beta(2)-adrenergic receptors (beta(2)-ARs) signal through Galpha(s) to activate the mitogen-activated protein kinase pathway in mouse embryonic fibroblast cells. At high agonist concentrations, signals are also transduced through beta(2)-ARs via an additional pathway that is G-protein-independent but tyrosine kinase Src-dependent. This new dosage-dependent switch of signaling modes of GPCRs has significant implications for GPCR intrinsic properties and desensitization.
Figures





References
-
- Ali MS, Sayeski PP, Dirksen LB, Hayzer DJ, Marrero MB, Bernstein KE (1997) Dependence on the motif YIPP for the physical association of Jak2 kinase with the intracellular carboxyl tail of the angiotensin II AT1 receptor. J Biol Chem 272: 23382–23388 - PubMed
-
- Bastepe M, Gunes Y, Perez-Villamil B, Hunzelman J, Weinstein LS, Juppner H (2002) Receptor-mediated adenylyl cyclase activation through XLalpha(s), the extra-large variant of the stimulatory G protein alpha-subunit. Mol Endocrinol 16: 1912–1919 - PubMed
-
- Bockaert J, Fagni L, Dumuis A, Marin P (2004) GPCR interacting proteins (GIP). Pharmacol Ther 103: 203–221 - PubMed
-
- Bouvier M (2001) Oligomerization of G-protein-coupled transmitter receptors. Nat Rev Neurosci 2: 274–286 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

