Proteasome substrate degradation requires association plus extended peptide
- PMID: 17170706
- PMCID: PMC1782366
- DOI: 10.1038/sj.emboj.7601476
Proteasome substrate degradation requires association plus extended peptide
Abstract
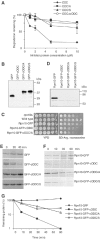
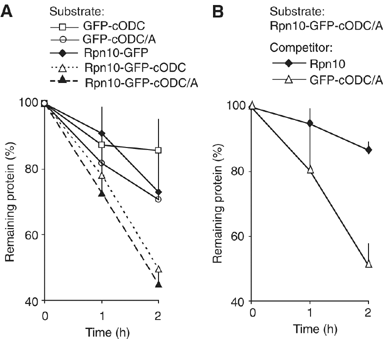
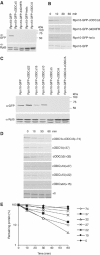
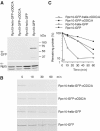
To determine the minimum requirements for substrate recognition and processing by proteasomes, the functional elements of a ubiquitin-independent degradation tag were dissected. The 37-residue C-terminus of ornithine decarboxylase (cODC) is a native degron, which also functions when appended to diverse proteins. Mutating the cysteine 441 residue within cODC impaired its proteasome association in the context of ornithine decarboxylase and prevented the turnover of GFP-cODC in yeast cells. Degradation of GFP-cODC with C441 mutations was restored by providing an alternate proteasome association element via fusion to the Rpn10 proteasome subunit. However, Rpn10-GFP was stable, unless extended by cODC or other peptides of similar size. In vitro reconstitution experiments confirmed the requirement for both proteasome tethering and a loosely structured region. Therefore, cODC and degradation tags in general must serve two functions: proteasome association and a site, consisting of an extended peptide region, used for initiating insertion into the protease.
Figures

References
-
- Carrion-Vazquez M, Oberhauser AF, Fisher TE, Marszalek PE, Li H, Fernandez JM (2000) Mechanical design of proteins studied by single-molecule force spectroscopy and protein engineering. Prog Biophys Mol Biol 74: 63–91 - PubMed
-
- Chen H, MacDonald A, Coffino P (2002) Structural elements of antizymes 1 and 2 required for proteasomal degradation of ornithine decarboxylase. J Biol Chem 277: 45957–45961 - PubMed
-
- Chen P, Johnson P, Sommer T, Jentsch S, Hochstrasser M (1993) Multiple ubiquitin-conjugating enzymes participate in the in vivo degradation of the yeast MATà2 repressor. Cell 74: 357–369 - PubMed
-
- Ghoda L, van Daalen Wetters T, Macrae M, Ascherman D, Coffino P (1989) Prevention of rapid intracellular degradation of ODC by a carboxyl-terminal truncation. Science 243: 1493–1495 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

