Cytoskeletal tension regulates both expression and degradation of h2-calponin in lung alveolar cells
- PMID: 17176089
- PMCID: PMC1764619
- DOI: 10.1021/bi061718f
Cytoskeletal tension regulates both expression and degradation of h2-calponin in lung alveolar cells
Abstract
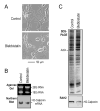
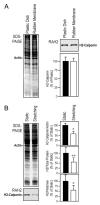
Calponin is an actin filament-associated regulatory protein, and its h2 isoform is expressed in lung alveolar epithelial cells under postnatal upregulation during lung development corresponding to the commencement of respiratory expansion. Consistent with this correlation to mechanical tension, the expression of h2-calponin in alveolar cells is dependent on substrate stiffness and cytoskeleton tension. The function of h2-calponin in the stability of actin cytoskeleton implicates a role in balancing the strength and compliance of alveoli. An interesting finding is a rapid degradation of h2-calponin in lung after prolonged deflation, which is prevented by inflation of the lung to the in situ expanded volume. Decreasing mechanical tension in cultured alveolar cells by reducing the dimension of culture matrix reproduced the degradation of h2-calponin. Inhibition of myosin II ATPase also resulted in the degradation of h2-calponin in alveolar cells, showing a determining role of the tension in the actin cytoskeleton. Alveolar cells statically cultured on silicon rubber membrane build high tension in the cytoskeleton corresponding to a high expression of h2-calponin. Chronic cyclic stretching of cells on the membrane did not increase but decreased the expression of h2-calponin. This finding suggests that when cellular structure adapts to the stretched dimension, cyclic relaxations periodically release cytoskeleton tension and lower the total amount of tension that the cell senses over time. Therefore, the isometric tension, other than tension dynamics, determines the expression of h2-calponin. The tension regulation of h2-calponin synthesis and degradation demonstrates a novel mechanical regulation of cellular biochemistry.
Figures






Similar articles
-
h2-Calponin is regulated by mechanical tension and modifies the function of actin cytoskeleton.J Biol Chem. 2005 Dec 23;280(51):42442-53. doi: 10.1074/jbc.M509952200. Epub 2005 Oct 18. J Biol Chem. 2005. PMID: 16236705 Free PMC article.
-
Mechanoregulation of SM22α/Transgelin.Biochemistry. 2017 Oct 17;56(41):5526-5538. doi: 10.1021/acs.biochem.7b00794. Epub 2017 Sep 27. Biochemistry. 2017. PMID: 28898058
-
Expression and purification of the h1 and h2 isoforms of calponin.Protein Expr Purif. 2003 Oct;31(2):231-9. doi: 10.1016/s1046-5928(03)00185-2. Protein Expr Purif. 2003. PMID: 14550641
-
Calponin in non-muscle cells.Cell Biochem Biophys. 2008;52(3):139-48. doi: 10.1007/s12013-008-9031-6. Epub 2008 Oct 23. Cell Biochem Biophys. 2008. PMID: 18946636 Review.
-
Calponin isoforms CNN1, CNN2 and CNN3: Regulators for actin cytoskeleton functions in smooth muscle and non-muscle cells.Gene. 2016 Jul 1;585(1):143-153. doi: 10.1016/j.gene.2016.02.040. Epub 2016 Mar 10. Gene. 2016. PMID: 26970176 Free PMC article. Review.
Cited by
-
Loss of Calponin 2 causes age-progressive proteinuria in mice.Physiol Rep. 2022 Sep;10(18):e15370. doi: 10.14814/phy2.15370. Physiol Rep. 2022. PMID: 36117313 Free PMC article.
-
Loss of Calponin 2 causes premature ovarian insufficiency in mice.J Ovarian Res. 2024 Feb 9;17(1):37. doi: 10.1186/s13048-024-01346-y. J Ovarian Res. 2024. PMID: 38336796 Free PMC article.
-
The Absence of Calponin 2 in Rabbits Suggests Caution in Choosing Animal Models.Front Bioeng Biotechnol. 2020 Feb 28;8:42. doi: 10.3389/fbioe.2020.00042. eCollection 2020. Front Bioeng Biotechnol. 2020. PMID: 32185166 Free PMC article.
-
Calponin 3 regulates actin cytoskeleton rearrangement in trophoblastic cell fusion.Mol Biol Cell. 2010 Nov 15;21(22):3973-84. doi: 10.1091/mbc.E10-03-0261. Epub 2010 Sep 22. Mol Biol Cell. 2010. PMID: 20861310 Free PMC article.
-
Downregulation of calponin 2 contributes to the quiescence of lung macrophages.Am J Physiol Cell Physiol. 2019 Oct 1;317(4):C749-C761. doi: 10.1152/ajpcell.00036.2019. Epub 2019 Jul 31. Am J Physiol Cell Physiol. 2019. PMID: 31365293 Free PMC article.
References
-
- Chicurel ME, Chen CS, Ingber DE. Cellular control lies in the balance of forces. Curr Opin Cell Biol. 1998;10:232–239. - PubMed
-
- Eckes B, Krieg T. Regulation of connective tissue homeostasis in the skin by mechanical forces. Clin Exp Rheumatol. 2004;22:S73–S76. - PubMed
-
- Hamill OP, Martinac B. Molecular basis of mechanotransduction in living cells. Physiol Rev. 2001;81:685–740. - PubMed
-
- Lehoux S, Tedgui A. Cellular mechanics and gene expression in blood vessels. J Biomech. 2003;36:631–643. - PubMed
-
- Walker JL, Fournier AK, Assoian RK. Regulation of growth factor signaling and cell cycle progression by cell adhesion and adhesion-dependent changes in cellular tension. Cytokine Growth Factor Rev. 2005;16:395–405. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

