Mapping the antigenicity of the parasites in Leishmania donovani infection by proteome serology
- PMID: 17183669
- PMCID: PMC1762392
- DOI: 10.1371/journal.pone.0000040
Mapping the antigenicity of the parasites in Leishmania donovani infection by proteome serology
Abstract
Background: Leishmaniasis defines a cluster of protozoal diseases with diverse clinical manifestations. The visceral form caused by Leishmania donovani is the most severe. So far, no vaccines exist for visceral leishmaniasis despite indications of naturally developing immunity, and sensitive immunodiagnostics are still at early stages of development.
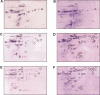
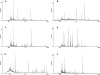
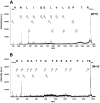
Methodology/principle findings: Establishing a proteome-serological methodology, we mapped the antigenicity of the parasites and the specificities of the immune responses in human leishmaniasis. Using 2-dimensional Western blot analyses with sera and parasites isolated from patients in India, we detected immune responses with widely divergent specificities for up to 330 different leishmanial antigens. 68 antigens were assigned to proteins in silver- and fluorochrome-stained gels. The antigenicity of these proteins did not correlate with the expression levels of the proteins. Although some antigens are shared among different parasite isolates, there are extensive differences and no immunodominant antigens, but indications of antigenic drift in the parasites. Six antigens were identified by mass spectrometry.
Conclusions/significance: Proteomics-based dissection of the serospecificities of leishmaniasis patients provides a comprehensive inventory of the complexity and interindividual heterogeneity of the host-responses to and variations in the antigenicity of the Leishmania parasites. This information can be instrumental in the development of vaccines and new immune monitoring and diagnostic devices.
Conflict of interest statement
Figures


Similar articles
-
Identification of new antigens in visceral leishmaniasis by expression cloning and immunoblotting with sera of kala-azar patients from Bihar, India.Infect Immun. 2005 Oct;73(10):7018-21. doi: 10.1128/IAI.73.10.7018-7021.2005. Infect Immun. 2005. PMID: 16177384 Free PMC article.
-
Prophylactic efficacy of high-molecular-weight antigenic fractions of a recent clinical isolate of Leishmania donovani against visceral leishmaniasis.Scand J Immunol. 2008 Nov;68(5):492-501. doi: 10.1111/j.1365-3083.2008.02171.x. Epub 2008 Sep 18. Scand J Immunol. 2008. PMID: 18803606
-
Serodiagnostic and immunoprophylactic potential of a 78kDa protein of Leishmania donovani of Indian origin.Med Sci Monit. 2002 Apr;8(4):BR117-22. Med Sci Monit. 2002. PMID: 11951057
-
Visceral leishmaniasis: host-parasite interactions and clinical presentation in the immunocompetent and in the immunocompromised host.Int J Infect Dis. 2013 Aug;17(8):e572-6. doi: 10.1016/j.ijid.2012.12.024. Epub 2013 Feb 4. Int J Infect Dis. 2013. PMID: 23380419 Review.
-
Immune responses in kala-azar.Indian J Med Res. 2006 Mar;123(3):245-66. Indian J Med Res. 2006. PMID: 16778308 Review.
Cited by
-
Heterogeneity of Leishmania donovani parasites complicates diagnosis of visceral leishmaniasis: comparison of different serological tests in three endemic regions.PLoS One. 2015 Mar 3;10(3):e0116408. doi: 10.1371/journal.pone.0116408. eCollection 2015. PLoS One. 2015. PMID: 25734336 Free PMC article.
-
The Leishmania infantum PUF proteins are targets of the humoral response during visceral leishmaniasis.BMC Res Notes. 2010 Jan 21;3:13. doi: 10.1186/1756-0500-3-13. BMC Res Notes. 2010. PMID: 20180988 Free PMC article.
-
Pathogenicity of Leishmania donovani is associated with the high expression of a group low molecular weight proteins.Trop Parasitol. 2015 Jul-Dec;5(2):106-17. doi: 10.4103/2229-5070.162521. Trop Parasitol. 2015. PMID: 26629453 Free PMC article.
-
Kinetoplastid genomics: the thin end of the wedge.Infect Genet Evol. 2008 Dec;8(6):901-6. doi: 10.1016/j.meegid.2008.07.001. Epub 2008 Jul 15. Infect Genet Evol. 2008. PMID: 18675383 Free PMC article.
-
rKLO8, a novel Leishmania donovani - derived recombinant immunodominant protein for sensitive detection of visceral leishmaniasis in Sudan.PLoS Negl Trop Dis. 2013 Jul 18;7(7):e2322. doi: 10.1371/journal.pntd.0002322. Print 2013. PLoS Negl Trop Dis. 2013. PMID: 23875052 Free PMC article.
References
-
- Chang KP, Dwyer DM. Multiplication of a human parasite (Leishmania donovani) in phagolysosomes of hamster macrophages in vitro. Science. 1976;193:678–680. - PubMed
-
- Killick-Kendrick R. The life-cycle of Leishmania in the sandfly with special reference to the form infective to the vertebrate host. Ann Parasitol Hum Comp. 1990;65(Suppl 1):37–42. - PubMed
-
- Pearson RD, Wheeler DA, Harrison LH, Kay HD. The immunobiology of leishmaniasis. Rev Infect Dis. 1983;5:907–927. - PubMed
-
- Davidson RN. Visceral leishmaniasis in clinical practice. J Infect. 1999;39:112–6. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

