Effects of glial cell line-derived neurotrophic factor deletion on ventral mesencephalic organotypic tissue cultures
- PMID: 17184739
- PMCID: PMC2670563
- DOI: 10.1016/j.brainres.2006.11.052
Effects of glial cell line-derived neurotrophic factor deletion on ventral mesencephalic organotypic tissue cultures
Abstract
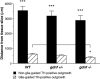
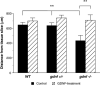
Glial cell line-derived neurotrophic factor (GDNF) is potent for survival and promotion of nerve fibers from midbrain dopamine neurons. It is also known to exert different effects on specific subpopulations of dopamine neurons. In organotypic tissue cultures, dopamine neurons form two diverse nerve fiber growth patterns, targeting the striatum differently. The aim of this study was to investigate the effect of GDNF on the formation of dopamine nerve fibers. Organotypic tissue cultures of ventral mesencephalon of gdnf gene-deleted mice were studied. The results revealed that dopamine neurons survive in the absence of GDNF. Tyrosine hydroxylase immunoreactivity demonstrated, in gdnf knockout and wildtype cultures, nerve fiber formation with two separate morphologies occurring either in the absence or the presence of astrocytes. The outgrowth that occurred in the absence of astrocytes was unaffected by gdnf deletion, whereas nerve fibers guided by the presence of astrocytes were affected in that they reached significantly shorter distances from the gdnf gene-deleted tissue slice, compared to those measured in wildtype cultures. Treatment with GDNF reversed this effect and increased nerve fiber density independent of genotype. Furthermore, migration of astrocytes reached significantly shorter distances from the tissue slice in GDNF knockout compared to wildtype cultures. Exogenous GDNF increased astrocytic migration in gdnf gene-deleted tissue cultures, comparable to lengths observed in wildtype tissue cultures. In conclusion, cultured midbrain dopamine neurons survive in the absence of GDNF, and the addition of GDNF improved dopamine nerve fiber formation - possibly as an indirect effect of astrocytic stimulation.
Figures





References
-
- Airavaara M, Planken A, Gäddnäs H, Piepponen TP, Saarma M, Ahtee L. Increased extracellular dopamine concentrations and FosB/DFosB expression in striatal brain areas of heterozygous GDNF knockout mice. Eur. J. Neurosci. 2004;20:2336–2344. - PubMed
-
- Apostolides C, Sanford E, Hong M, Medez I. Glial cell line-derived neurotrophic factor improves intrastriatal graft survival of stored dopaminergic cells. Neuroscience. 1998;83:363–372. - PubMed
-
- Barker RA, Dunnett SB, Faissner A, Fawcett JW. The time course of loss of dopaminergic neurons and the gliotic reaction surrounding grafts of embryonic mesencephalon to the striatum. Exp. Neurol. 1996;141:79–93. - PubMed
-
- Barroso-Chinea P, Cruz-Muros I, Aymerich MS, Rodríguez-Díaz ,M, Afonso-Oramas D, Lanciego JL, González-Hernández ,T. Striatal expression of GDNF and different vulnerability of midbrain dopaminergic neurons. Eur. J. Neurosci. 2005;21:1527–1815. - PubMed
-
- Beck KD, Valverde J, Alexi T, Poulsen K, Moffat B, Vandlen RA, Rosenthal A, Hefti F. Mesencephalic dopaminergic neurons protected by GDNF from axotomy-induced degeneration in the adult brain. Nature. 1995;373:339–341. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

