Regulation of systemic and local neutrophil responses by G-CSF during pulmonary Pseudomonas aeruginosa infection
- PMID: 17185469
- PMCID: PMC1852251
- DOI: 10.1182/blood-2005-01-015081
Regulation of systemic and local neutrophil responses by G-CSF during pulmonary Pseudomonas aeruginosa infection
Abstract
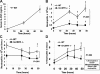
Granulocyte colony-stimulating factor (G-CSF) regulates the production, maturation, and function of neutrophils. Its expression is often induced during infection, resulting in high concentrations of G-CSF in inflammatory exudates and in the blood, suggesting that it may regulate both local and systemic neutrophil responses. Herein, we characterize the neutrophil response in G-CSFR(-/-) mice following intratracheal injection with Pseudomonas aeruginosa-laden agarose beads, modeling the pulmonary infection observed in many patients with cystic fibrosis. G-CSFR(-/-) mice are markedly susceptible to bronchopulmonary P aeruginosa infection, exhibiting decreased survival and bacterial clearance as well as extensive damage to lung tissue. The systemic neutrophil response was mediated primarily by enhanced neutrophil release from the bone marrow rather than increased neutrophil production and was attenuated in G-CSFR(-/-) mice. Despite normal to increased local production of inflammatory chemokines, neutrophil accumulation into the infected lung of G-CSFR(-/-) mice was markedly reduced. Moreover, the percentage of apoptotic neutrophils in the lung was elevated, suggesting that G-CSF signals may play an important role in regulating neutrophil survival at the inflammatory site. Collectively, these data provide new evidence that G-CSF signals play important but specific roles in the regulation of the systemic and local neutrophil response following infection.
Figures

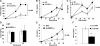
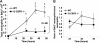


Similar articles
-
Neutrophil-Expressed p21/waf1 Favors Inflammation Resolution in Pseudomonas aeruginosa Infection.Am J Respir Cell Mol Biol. 2016 May;54(5):740-50. doi: 10.1165/rcmb.2015-0047OC. Am J Respir Cell Mol Biol. 2016. PMID: 26517580
-
Murine models of chronic Pseudomonas aeruginosa lung infection.Lab Anim. 2002 Jul;36(3):291-312. doi: 10.1258/002367702320162405. Lab Anim. 2002. PMID: 12144741
-
Role of G-CSF in monophosphoryl lipid A-mediated augmentation of neutrophil functions after burn injury.J Leukoc Biol. 2016 Apr;99(4):629-40. doi: 10.1189/jlb.4A0815-362R. Epub 2015 Nov 4. J Leukoc Biol. 2016. PMID: 26538529 Free PMC article.
-
Harnessing Neutrophil Survival Mechanisms during Chronic Infection by Pseudomonas aeruginosa: Novel Therapeutic Targets to Dampen Inflammation in Cystic Fibrosis.Front Cell Infect Microbiol. 2017 Jun 30;7:243. doi: 10.3389/fcimb.2017.00243. eCollection 2017. Front Cell Infect Microbiol. 2017. PMID: 28713772 Free PMC article. Review.
-
G-CSF - A double edge sword in neutrophil mediated immunity.Semin Immunol. 2021 Apr;54:101516. doi: 10.1016/j.smim.2021.101516. Epub 2021 Oct 30. Semin Immunol. 2021. PMID: 34728120 Review.
Cited by
-
Ethanol Intoxication Impairs Respiratory Function and Bacterial Clearance and Is Associated With Neutrophil Accumulation in the Lung After Streptococcus pneumoniae Infection.Front Immunol. 2022 May 4;13:884719. doi: 10.3389/fimmu.2022.884719. eCollection 2022. Front Immunol. 2022. PMID: 35603143 Free PMC article.
-
Leptin receptor q223r polymorphism influences neutrophil mobilization after Clostridium difficile infection.Mucosal Immunol. 2018 May;11(3):947-957. doi: 10.1038/mi.2017.119. Epub 2018 Jan 24. Mucosal Immunol. 2018. PMID: 29363668 Free PMC article.
-
Acidosis exacerbates in vivo IL-1-dependent inflammatory responses and neutrophil recruitment during pulmonary Pseudomonas aeruginosa infection.Am J Physiol Lung Cell Mol Physiol. 2018 Feb 1;314(2):L225-L235. doi: 10.1152/ajplung.00338.2017. Epub 2017 Oct 6. Am J Physiol Lung Cell Mol Physiol. 2018. PMID: 28982735 Free PMC article.
-
Comparative transcriptome analyses of Pseudomonas aeruginosa.Hum Genomics. 2009 Jul;3(4):349-61. doi: 10.1186/1479-7364-3-4-361. Hum Genomics. 2009. PMID: 19706365 Free PMC article. Review.
-
MSCs alleviate LPS-induced acute lung injury by inhibiting the proinflammatory function of macrophages in mouse lung organoid-macrophage model.Cell Mol Life Sci. 2024 Mar 11;81(1):124. doi: 10.1007/s00018-024-05150-1. Cell Mol Life Sci. 2024. PMID: 38466420 Free PMC article.
References
-
- Carulli G. Effects of recombinant human granulocyte colony-stimulating factor administration on neutrophil phenotype and functions. Haematologica. 1997;82:606–616. - PubMed
-
- Anderlini P, Przepiorka D, Champlin R, Korbling M. Biologic and clinical effects of granulocyte colony-stimulating factor in normal individuals. Blood. 1996;88:2819–2825. - PubMed
-
- Basu S, Dunn A, Ward A. G-CSF: function and modes of action (review). Int J Mol Med. 2002;10:3–10. - PubMed
-
- Lieschke GJ, Grail D, Hodgson G, et al. Mice lacking granulocyte colony-stimulating factor have chronic neutropenia, granulocyte and macrophage progenitor cell deficiency, and impaired neutrophil mobilization. Blood. 1994;84:1737–1746. - PubMed
-
- Liu F, Wu HY, Wesselschmidt R, Kornaga T, Link DC. Impaired production and increased apoptosis of neutrophils in granulocyte colony-stimulating factor receptor-deficient mice. Immunity. 1996;5:491–501. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

