Structure and quantum chemical characterization of chloroperoxidase compound 0, a common reaction intermediate of diverse heme enzymes
- PMID: 17190816
- PMCID: PMC1765485
- DOI: 10.1073/pnas.0606285103
Structure and quantum chemical characterization of chloroperoxidase compound 0, a common reaction intermediate of diverse heme enzymes
Abstract
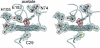
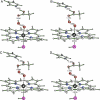
We have determined the crystal structure of the chloroperoxidase (CPO) hydroperoxo reaction intermediate (CPO compound 0) at 1.75-A resolution. The intermediate was generated through controlled photoreduction of the CPO oxygen complex during x-ray data collection, which was monitored by recording of the crystal absorption spectra. Initially, the peroxo-anion species was formed and then protonated to yield compound 0. Quantum chemical calculations indicate that the peroxo-anion species is not stable and collapses instantaneously to compound 0. Compound 0 is present in the ferric low-spin doublet ground state and is characterized by a long O O bond length of 1.5 A and a Fe O bond distance of 1.8 A, which is also observed in the crystal structure.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Quantum mechanical/molecular mechanical study on the mechanisms of compound I formation in the catalytic cycle of chloroperoxidase: an overview on heme enzymes.J Phys Chem B. 2008 Aug 7;112(31):9490-500. doi: 10.1021/jp803010f. Epub 2008 Jul 3. J Phys Chem B. 2008. PMID: 18597525
-
The ferric-hydroperoxo complex of chloroperoxidase.Biochem Biophys Res Commun. 2007 Nov 30;363(4):954-8. doi: 10.1016/j.bbrc.2007.09.085. Epub 2007 Oct 1. Biochem Biophys Res Commun. 2007. PMID: 17920039 Free PMC article.
-
The second step of the nitric oxide synthase reaction: evidence for ferric-peroxo as the active oxidant.J Am Chem Soc. 2009 Jan 14;131(1):297-305. doi: 10.1021/ja807299t. J Am Chem Soc. 2009. PMID: 19128180
-
Crystal structures of chloroperoxidase with its bound substrates and complexed with formate, acetate, and nitrate.J Biol Chem. 2006 Aug 18;281(33):23990-8. doi: 10.1074/jbc.M603166200. Epub 2006 Jun 20. J Biol Chem. 2006. PMID: 16790441
-
Stereochemistry of the chloroperoxidase active site: crystallographic and molecular-modeling studies.Chem Biol. 1998 Sep;5(9):461-73. doi: 10.1016/s1074-5521(98)90003-5. Chem Biol. 1998. PMID: 9751642
Cited by
-
Radiation damage in macromolecular crystallography: what is it and why should we care?Acta Crystallogr D Biol Crystallogr. 2010 Apr;66(Pt 4):339-51. doi: 10.1107/S0907444910008656. Epub 2010 Mar 24. Acta Crystallogr D Biol Crystallogr. 2010. PMID: 20382986 Free PMC article.
-
Isolating Fe-O2 Intermediates in Dioxygen Activation by Iron Porphyrin Complexes.Molecules. 2022 Jul 22;27(15):4690. doi: 10.3390/molecules27154690. Molecules. 2022. PMID: 35897870 Free PMC article. Review.
-
Electron paramagnetic resonance and electron-nuclear double resonance studies of the reactions of cryogenerated hydroperoxoferric-hemoprotein intermediates.Biochemistry. 2014 Aug 5;53(30):4894-903. doi: 10.1021/bi500296d. Epub 2014 Jul 21. Biochemistry. 2014. PMID: 25046203 Free PMC article.
-
Lessons on O2 and NO bonding to heme from ab initio multireference/multiconfiguration and DFT calculations.J Biol Inorg Chem. 2011 Aug;16(6):841-55. doi: 10.1007/s00775-011-0763-1. Epub 2011 Mar 4. J Biol Inorg Chem. 2011. PMID: 21373960
-
Characterization of a thiolato iron(III) Peroxy dianion complex.Angew Chem Int Ed Engl. 2012 Sep 3;51(36):9132-6. doi: 10.1002/anie.201203602. Epub 2012 Aug 6. Angew Chem Int Ed Engl. 2012. PMID: 22888066 Free PMC article.
References
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources

