Identification and characterization of DEDDL, a human-specific isoform of DEDD
- PMID: 17193921
- PMCID: PMC6032443
- DOI: 10.3727/000000006783991836
Identification and characterization of DEDDL, a human-specific isoform of DEDD
Abstract
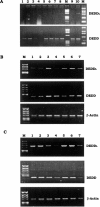
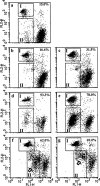
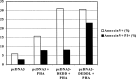
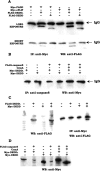
Death effector domain (DED) containing molecules are usually involved in the intracellular apoptosis cascade as executioners or regulators. One of these molecules, DEDD, was identified as a final target of the CD95 signaling pathway by which it would be transferred into the nucleolus to inhibit RNA polymerase I-dependent transcription. Here we describe a longer isoform of DEDD, DEDDL, produced by alternatively splicing, as an immune cell-specific DED-containing molecule. It is only expressed in human T lymphocytes and dendritic cells (DCs), and the mRNA expression in DCs was elevated upon inductive maturation. In cell lines MCF-7 and Jurkat, the overexpression of DEDDL could induce apoptosis more potently than that of DEDD. That DEDDL could bind FADD and cFLIP more potently than DEDD in vivo was revealed by cotransfection and immunoprecipitation. This may explain why DEDDL is a more potent apoptosis inducer, because DED-containing proteins usually induce apoptosis through DED binding. Finally, why DEDD and DEDDL are unstable in the overexpression and other studies may be explained by the finding that they are potential substrates of active caspases.
Figures

Similar articles
-
DEDD, a novel death effector domain-containing protein, targeted to the nucleolus.EMBO J. 1998 Oct 15;17(20):5974-86. doi: 10.1093/emboj/17.20.5974. EMBO J. 1998. PMID: 9774341 Free PMC article.
-
DEDD and DEDD2 associate with caspase-8/10 and signal cell death.Oncogene. 2003 Jan 16;22(2):291-7. doi: 10.1038/sj.onc.1206099. Oncogene. 2003. PMID: 12527898
-
Nuclear localization of DEDD leads to caspase-6 activation through its death effector domain and inhibition of RNA polymerase I dependent transcription.Cell Death Differ. 2001 Dec;8(12):1157-68. doi: 10.1038/sj.cdd.4400928. Cell Death Differ. 2001. PMID: 11753564
-
Death effector domain-containing proteins.Cell Mol Life Sci. 2009 Mar;66(5):814-30. doi: 10.1007/s00018-008-8489-0. Cell Mol Life Sci. 2009. PMID: 18989622 Free PMC article. Review.
-
The death effector domain protein family: regulators of cellular homeostasis.Nat Immunol. 2003 May;4(5):404-9. doi: 10.1038/ni0503-404. Nat Immunol. 2003. PMID: 12719729 Review.
References
-
- Aravind L.; Dixit V. M.; Koonin E. V. Apoptotic molecular machinery: Vastly increased complexity in vertebrates revealed by genome comparisons. Science 291:1279–1284; 2001. - PubMed
-
- Ashany D.; Savir A.; Bhardwaj N.; Elkon K. B. Dendritic cells are resistant to apoptosis through the Fas (CD95/APO-1) pathway. J. Immunol. 163:5303–5311; 1999. - PubMed
-
- Ashkenazi A.; Dixit V. M. Death receptors: Signaling and modulation. Science 281:1305–1308; 1998. - PubMed
-
- Barnhart B. C.; Lee J. C.; Alappat E. C.; Peter M. E. The death effector domain protein family. Oncogene 22:8634–8644; 2003. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
