Timing and sequence of brain activity in top-down control of visual-spatial attention
- PMID: 17199410
- PMCID: PMC1761048
- DOI: 10.1371/journal.pbio.0050012
Timing and sequence of brain activity in top-down control of visual-spatial attention
Abstract
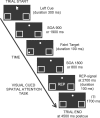
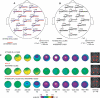
Recent brain imaging studies using functional magnetic resonance imaging (fMRI) have implicated a frontal-parietal network in the top-down control of attention. However, little is known about the timing and sequence of activations within this network. To investigate these timing questions, we used event-related electrical brain potentials (ERPs) and a specially designed visual-spatial attentional-cueing paradigm, which were applied as part of a multi-methodological approach that included a closely corresponding event-related fMRI study using an identical paradigm. In the first 400 ms post cue, attention-directing and control cues elicited similar general cue-processing activity, corresponding to the more lateral subregions of the frontal-parietal network identified with the fMRI. Following this, the attention-directing cues elicited a sustained negative-polarity brain wave that was absent for control cues. This activity could be linked to the more medial frontal-parietal subregions similarly identified in the fMRI as specifically involved in attentional orienting. Critically, both the scalp ERPs and the fMRI-seeded source modeling for this orienting-related activity indicated an earlier onset of frontal versus parietal contribution ( approximately 400 versus approximately 700 ms). This was then followed ( approximately 800-900 ms) by pretarget biasing activity in the region-specific visual-sensory occipital cortex. These results indicate an activation sequence of key components of the attentional-control brain network, providing insight into their functional roles. More specifically, these results suggest that voluntary attentional orienting is initiated by medial portions of frontal cortex, which then recruit medial parietal areas. Together, these areas then implement biasing of region-specific visual-sensory cortex to facilitate the processing of upcoming visual stimuli.
Conflict of interest statement
Competing interests. The authors have declared that no competing interests exist.
Figures



Comment in
-
The neural cascade of attentional control.PLoS Biol. 2007 Jan;5(1):e27. doi: 10.1371/journal.pbio.0050027. Epub 2007 Jan 2. PLoS Biol. 2007. PMID: 20076645 Free PMC article. No abstract available.
References
-
- Posner MI, Snyder CR, Davidson BJ. Attention and the detection of signals. J Exp Psychol. 1980;109:160–174. - PubMed
-
- Downing CJ. Expectancy and visual-spatial attention: Effects on perceptual quality. J Exp Psychol Hum Percept Perform. 1988;14:188–202. - PubMed
-
- Mangun GR, Hillyard SA. Modulations of sensory-evoked brain potentials indicate changes in perceptual processing during visual-spatial priming. J Exp Psychol Hum Percept Perform. 1991;17:1057–1074. - PubMed
-
- Heinze HJ, Mangun GR, Burchert W, Hinrichs H, Scholz M, et al. Combined spatial and temporal imaging of brain activity during visual selective attention in humans. Nature. 1994;372:543–546. - PubMed
-
- Woldorff MG, Fox PT, Matzke M, Lancaster JL, Veeraswamy S, et al. Retinotopic organization of early visual spatial attention effects as revealed by PET and ERPs. Human Brain Mapping. 1997;5:280–286. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

