Adrenomedullin protects from experimental arthritis by down-regulating inflammation and Th1 response and inducing regulatory T cells
- PMID: 17200199
- PMCID: PMC1762686
- DOI: 10.2353/ajpath.2007.060596
Adrenomedullin protects from experimental arthritis by down-regulating inflammation and Th1 response and inducing regulatory T cells
Abstract
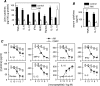
Rheumatoid arthritis is a chronic autoimmune disease of unknown etiology characterized by chronic inflammation in the joints and subsequent destruction of the cartilage and bone. The present study proposes a new strategy for the treatment of arthritis: the administration of the immunomodulatory neuropeptide adrenomedullin. Treatment with adrenomedullin significantly reduced incidence and severity of collagen-induced arthritis, an experimental model of rheumatoid arthritis, completely abrogating joint swelling and destruction of cartilage and bone. The therapeutic effect of adrenomedullin was associated with a striking reduction of the two deleterious components of the disease, ie, the Th1-driven autoimmune and inflammatory responses. Adrenomedullin also induced the generation and/or activation of efficient CD4+ CD25+ regulatory T cells in arthritis with capacity to suppress autoreactive response and restore immune tolerance, which could play a pivotal role in the therapeutic effect of adrenomedullin on experimental arthritis contributing to the restoration of immune tolerance.
Figures



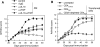
Similar articles
-
Therapeutic effect of urocortin on collagen-induced arthritis by down-regulation of inflammatory and Th1 responses and induction of regulatory T cells.Arthritis Rheum. 2007 Feb;56(2):531-43. doi: 10.1002/art.22394. Arthritis Rheum. 2007. PMID: 17265488
-
Treatment of experimental arthritis by inducing immune tolerance with human adipose-derived mesenchymal stem cells.Arthritis Rheum. 2009 Apr;60(4):1006-19. doi: 10.1002/art.24405. Arthritis Rheum. 2009. PMID: 19333946
-
Vasoactive intestinal peptide prevents experimental arthritis by downregulating both autoimmune and inflammatory components of the disease.Nat Med. 2001 May;7(5):563-8. doi: 10.1038/87887. Nat Med. 2001. PMID: 11329057
-
The role of the T cell in autoimmune inflammation.Arthritis Res Ther. 2005;7 Suppl 2(Suppl 2):S4-14. doi: 10.1186/ar1703. Epub 2005 Mar 16. Arthritis Res Ther. 2005. PMID: 15833146 Free PMC article. Review.
-
Cortistatin as a potential multistep therapeutic agent for inflammatory disorders.Drug News Perspect. 2006 Sep;19(7):393-9. doi: 10.1358/dnp.2006.19.7.1021490. Drug News Perspect. 2006. PMID: 17080202 Review.
Cited by
-
Role of adrenomedullin in Lyme disease.Infect Immun. 2010 Dec;78(12):5307-13. doi: 10.1128/IAI.00630-10. Epub 2010 Oct 4. Infect Immun. 2010. PMID: 20921145 Free PMC article.
-
Adrenomedullin, a Novel Target for Neurodegenerative Diseases.Mol Neurobiol. 2018 Dec;55(12):8799-8814. doi: 10.1007/s12035-018-1031-y. Epub 2018 Mar 29. Mol Neurobiol. 2018. PMID: 29600350 Review.
-
Adrenomedullin Regulates IL-1β Gene Expression in F4/80+ Macrophages during Synovial Inflammation.J Immunol Res. 2017;2017:9832430. doi: 10.1155/2017/9832430. Epub 2017 Feb 19. J Immunol Res. 2017. PMID: 28299347 Free PMC article.
-
Anti-inflammatory effects of adrenomedullin on acute lung injury induced by Carrageenan in mice.Mediators Inflamm. 2012;2012:717851. doi: 10.1155/2012/717851. Epub 2012 May 20. Mediators Inflamm. 2012. PMID: 22685374 Free PMC article.
-
An RNA vaccine against adrenomedullin reduces angiogenesis and tumor burden in a syngeneic metastatic melanoma mouse model.Front Immunol. 2025 Jun 5;16:1604156. doi: 10.3389/fimmu.2025.1604156. eCollection 2025. Front Immunol. 2025. PMID: 40539045 Free PMC article.
References
-
- Anthony DD, Haqqi TM. Collagen-induced arthritis in mice: an animal model to study the pathogenesis of rheumatoid arthritis. Clin Exp Rheumatol. 1999;17:240–244. - PubMed
-
- Feldmann M, Brennan FM, Maini RN. Role of cytokines in rheumatoid arthritis. Annu Rev Immunol. 1996;14:397–440. - PubMed
-
- Brand DD, Kang AH, Rosloniec EF. Immunopathogenesis of collagen arthritis. Springer Semin Immunopathol. 2003;25:3–18. - PubMed
-
- Feldmann M, Maini RN. Anti-TNF alpha therapy of rheumatoid arthritis: what have we learned? Annu Rev Immunol. 2001;19:163–196. - PubMed
-
- Hinson JP, Kapas S, Smith DM. Adrenomedullin, a multifunctional regulatory peptide. Endocr Rev. 2000;21:138–167. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

