Human cytomegalovirus UL38 protein blocks apoptosis
- PMID: 17202209
- PMCID: PMC1866066
- DOI: 10.1128/JVI.02124-06
Human cytomegalovirus UL38 protein blocks apoptosis
Abstract
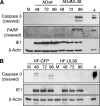
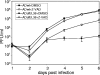
Apoptosis is an innate cellular defense response to viral infection. The slow-replicating human cytomegalovirus (HCMV) blocks premature death of host cells prior to completion of the infection cycle. In this study, we report that the HCMV UL38 gene encodes a cell death inhibitory protein. A mutant virus lacking the pUL38 coding sequence, ADdlUL38, grew poorly in human fibroblasts, failed to accumulate viral DNA to wild-type levels, and induced excessive death of infected cells. Cells expressing pUL38 were resistant to cell death upon infection and effectively supported the growth of ADdlUL38. Cells infected with the pUL38-deficient virus showed morphological changes characteristic of apoptosis, including cell shrinkage, membrane blebbing, vesicle release, and chromatin condensation and fragmentation. The proteolytic cleavage of two key enzymes involved in apoptosis, namely, caspase 3 and poly(ADP-ribose) polymerase, was activated upon ADdlUL38 infection, and the cleavage was blocked in cells expressing pUL38. The pan-caspase inhibitor Z-VAD-FMK largely restored the growth of ADdlUL38 in normal fibroblasts, indicating that the defective growth of the mutant virus mainly resulted from premature death of host cells. Furthermore, cells expressing pUL38 were resistant to cell death induced by a mutant adenovirus lacking the antiapoptotic E1B-19K protein or by thapsigargin, which disrupts calcium homeostasis in the endoplasmic reticulum. Taken together, these results indicate that the HCMV protein pUL38 suppresses apoptosis, blocking premature death of host cells to facilitate efficient virus replication.
Figures







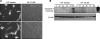
Similar articles
-
Human Cytomegalovirus Protein pUL38 Prevents Premature Cell Death by Binding to Ubiquitin-Specific Protease 24 and Regulating Iron Metabolism.J Virol. 2018 Jun 13;92(13):e00191-18. doi: 10.1128/JVI.00191-18. Print 2018 Jul 1. J Virol. 2018. PMID: 29695420 Free PMC article.
-
Tuberous Sclerosis Complex Protein 2-Independent Activation of mTORC1 by Human Cytomegalovirus pUL38.J Virol. 2015 Aug;89(15):7625-35. doi: 10.1128/JVI.01027-15. Epub 2015 May 13. J Virol. 2015. PMID: 25972538 Free PMC article.
-
Human cytomegalovirus protein pUL38 induces ATF4 expression, inhibits persistent JNK phosphorylation, and suppresses endoplasmic reticulum stress-induced cell death.J Virol. 2009 Apr;83(8):3463-74. doi: 10.1128/JVI.02307-08. Epub 2009 Feb 4. J Virol. 2009. PMID: 19193809 Free PMC article.
-
The human cytomegalovirus protein pUL38 suppresses endoplasmic reticulum stress-mediated cell death independently of its ability to induce mTORC1 activation.J Virol. 2011 Sep;85(17):9103-13. doi: 10.1128/JVI.00572-11. Epub 2011 Jun 29. J Virol. 2011. PMID: 21715486 Free PMC article.
-
Control of apoptosis by human cytomegalovirus.Curr Top Microbiol Immunol. 2008;325:281-95. doi: 10.1007/978-3-540-77349-8_16. Curr Top Microbiol Immunol. 2008. PMID: 18637512 Review.
Cited by
-
US28 is a potent activator of phospholipase C during HCMV infection of clinically relevant target cells.PLoS One. 2012;7(11):e50524. doi: 10.1371/journal.pone.0050524. Epub 2012 Nov 29. PLoS One. 2012. PMID: 23209769 Free PMC article.
-
HtrA2/Omi terminates cytomegalovirus infection and is controlled by the viral mitochondrial inhibitor of apoptosis (vMIA).PLoS Pathog. 2008 May 9;4(5):e1000063. doi: 10.1371/journal.ppat.1000063. PLoS Pathog. 2008. PMID: 18769594 Free PMC article.
-
Global reprogramming of the cellular translational landscape facilitates cytomegalovirus replication.Cell Rep. 2014 Jan 16;6(1):9-17. doi: 10.1016/j.celrep.2013.11.045. Epub 2013 Dec 27. Cell Rep. 2014. PMID: 24373965 Free PMC article.
-
Human cytomegalovirus pUL79 is an elongation factor of RNA polymerase II for viral gene transcription.PLoS Pathog. 2014 Aug 28;10(8):e1004350. doi: 10.1371/journal.ppat.1004350. eCollection 2014 Aug. PLoS Pathog. 2014. PMID: 25166009 Free PMC article.
-
Molecular Insights into HR-HPV and HCMV Co-Presence in Cervical Cancer Development.Cancers (Basel). 2025 Feb 8;17(4):582. doi: 10.3390/cancers17040582. Cancers (Basel). 2025. PMID: 40002177 Free PMC article. Review.
References
-
- Adair, R., G. W. Liebisch, and A. M. Colberg-Poley. 2003. Complex alternative processing of human cytomegalovirus UL37 pre-mRNA. J. Gen. Virol. 84:3353-3358. - PubMed
-
- Andoniou, C. E., and M. A. Degli-Esposti. 2006. Insights into the mechanisms of CMV-mediated interference with cellular apoptosis. Immunol. Cell Biol. 84:99-106. - PubMed
-
- Arnoult, D., L. M. Bartle, A. Skaletskaya, D. Poncet, N. Zamzami, P. U. Park, J. Sharpe, R. J. Youle, and V. S. Goldmacher. 2004. Cytomegalovirus cell death suppressor vMIA blocks Bax- but not Bak-mediated apoptosis by binding and sequestering Bax at mitochondria. Proc. Natl. Acad. Sci. USA 101:7988-7993. - PMC - PubMed
-
- Benedict, C. A., P. S. Norris, and C. F. Ware. 2002. To kill or be killed: viral evasion of apoptosis. Nat. Immunol. 3:1013-1018. - PubMed
-
- Billstrom Schroeder, M., R. Christensen, and G. S. Worthen. 2002. Human cytomegalovirus protects endothelial cells from apoptosis induced by growth factor withdrawal. J. Clin. Virol. 25(Suppl. 2):S149-S157. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

