Lymphopenic mice reconstituted with limited repertoire T cells develop severe, multiorgan, Th2-associated inflammatory disease
- PMID: 17202252
- PMCID: PMC1761908
- DOI: 10.1073/pnas.0610289104
Lymphopenic mice reconstituted with limited repertoire T cells develop severe, multiorgan, Th2-associated inflammatory disease
Abstract
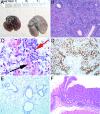
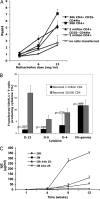
Lymphopenia and restricted T cell repertoires in humans are often associated with severe eosinophilic disease and a T cell Th2 bias. To examine the pathogenesis of this phenomenon, C57BL/6 Rag2-/- mice received limited (3 x 10(4)) or large (2 x 10(6)) numbers of CD4 T cells. Three to 5 months after transfer, mice that had received 3 x 10(4) T cells, but not those that received 2 x 10(6), developed fulminant macrophage pneumonia with eosinophilia, Ym1 deposition, and methacholine-induced airway hyperresponsiveness, as well as eosinophilic gastritis; esophagitis and other organ damage occurred in some cases. Donor cells were enriched for IL-4, IL-5, and IL-13 producers. When 3 x 10(4) cells were transferred into CD3epsilon-/- hosts, the mice developed strikingly elevated serum IgE. Prior transfer of 3 x 10(5) CD25+ CD4 T cells into Rag2-/- recipients prevented disease upon subsequent transfer of CD25- CD4 T cells, whereas 3 x 10(4) regulatory T cells (Tregs) did not, despite the fact that there were equal total numbers of Tregs in the host at the time of transfer of CD25- CD4 T cells. Limited repertoire complexity of Tregs may lead to a failure to control induction of immunopathologic responses, and limitation in repertoire complexity of conventional cells may be responsible for the Th2 phenotype.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Markert ML, Alexieff MJ, Li J, Sarzotti M, Ozaki DA, Devlin BH, Sempowski GD, Rhein ME, Szabolcs P, Hale LP, et al. J Allergy Clin Immunol. 2000;113:734–741. - PubMed
-
- Villa A, Santagata S, Bozzi F, Giliani S, Frattini A, Imberti L, Gatta LB, Ochs HD, Schwarz K, Notarangelo LD, et al. Cell. 1998;93:885–896. - PubMed
-
- Tietz A, Sponagel L, Erb P, Bucher H, Battegay M, Zimmerli W. Eur J Clin Microbiol Infect Dis. 1997;16:675–677. - PubMed
-
- Yamanaka K, Yawalkar N, Jones DA, Hurwitz D, Ferenczi K, Eapen S, Kupper TS. Clin Cancer Res. 2005;11:5748–5755. - PubMed
-
- Yawalkar N, Ferenczi K, Jones DA, Yamanaka K, Suh KY, Sadat S, Kupper TS. Blood. 2003;102:4059–4066. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

