RalA and RalB function as the critical GTP sensors for GTP-dependent exocytosis
- PMID: 17202486
- PMCID: PMC6672288
- DOI: 10.1523/JNEUROSCI.2537-06.2007
RalA and RalB function as the critical GTP sensors for GTP-dependent exocytosis
Abstract
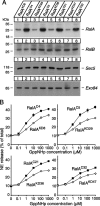
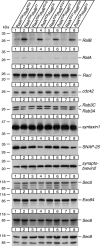
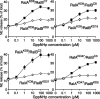
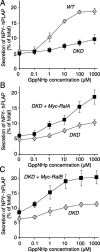
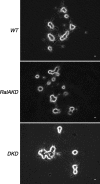
Although it has been established that the activation of GTPases by non-hydrolyzable GTP stimulates neurotransmitter release from many different secretory cell types, the underlying mechanisms remain unclear. In the present study we aimed to elucidate the functional role(s) for endogenous Ras-like protein A (RalA) and RalB GTPases in GTP-dependent exocytosis. For this purpose stable neuroendocrine pheochromocytoma 12 (PC12) cell lines were generated in which the expressions of both RalA and RalB were strongly downregulated. In these double knock-down cells GTP-dependent exocytosis was reduced severely and was restored after the expression of RalA or RalB was reintroduced by transfection. In contrast, Ca2+-dependent exocytosis and the docking of dense core vesicles analyzed by electron microscopy remained unchanged in the double knock-down cells. Furthermore, the transfected RalA and RalB appeared to be localized primarily on the dense core vesicles in undifferentiated and nerve growth factor-differentiated PC12 cells. Our results indicate that endogenous RalA and RalB function specifically as GTP sensors for the GTP-dependent exocytosis of dense core vesicles, but they are not required for the general secretory pathways, including tethering of vesicles to the plasma membrane and Ca2+-dependent exocytosis.
Figures





Similar articles
-
RalA-exocyst interaction mediates GTP-dependent exocytosis.J Biol Chem. 2004 May 7;279(19):19875-81. doi: 10.1074/jbc.M400522200. Epub 2004 Feb 20. J Biol Chem. 2004. PMID: 14978027
-
The Small GTPase RalA controls exocytosis of large dense core secretory granules by interacting with ARF6-dependent phospholipase D1.J Biol Chem. 2005 Aug 19;280(33):29921-8. doi: 10.1074/jbc.M413748200. Epub 2005 Jun 24. J Biol Chem. 2005. PMID: 15980073
-
RalB uncoupling from exocyst is required for endothelial Weibel-Palade body exocytosis.Mol Biol Cell. 2025 May 1;36(5):ar62. doi: 10.1091/mbc.E24-11-0493. Epub 2025 Apr 2. Mol Biol Cell. 2025. PMID: 40172988
-
Ral GTPases: crucial mediators of exocytosis and tumourigenesis.J Biochem. 2015 May;157(5):285-99. doi: 10.1093/jb/mvv029. Epub 2015 Mar 20. J Biochem. 2015. PMID: 25796063 Review.
-
The RAL Enigma: Distinct Roles of RALA and RALB in Cancer.Cells. 2022 May 14;11(10):1645. doi: 10.3390/cells11101645. Cells. 2022. PMID: 35626682 Free PMC article. Review.
Cited by
-
Vacuolar H(+)-ATPase subunits Voa1 and Voa2 cooperatively regulate secretory vesicle acidification, transmitter uptake, and storage.Mol Biol Cell. 2011 Sep;22(18):3394-409. doi: 10.1091/mbc.E11-02-0155. Epub 2011 Jul 27. Mol Biol Cell. 2011. PMID: 21795392 Free PMC article.
-
The GTPase RalA regulates different steps of the secretory process in pancreatic beta-cells.PLoS One. 2009 Nov 5;4(11):e7770. doi: 10.1371/journal.pone.0007770. PLoS One. 2009. PMID: 19890390 Free PMC article.
-
Ral function in muscle is required for flight maintenance in Drosophila.Small GTPases. 2020 May;11(3):174-179. doi: 10.1080/21541248.2017.1367456. Epub 2017 Dec 28. Small GTPases. 2020. PMID: 29284321 Free PMC article.
-
Different metastasis promotive potency of small G-proteins RalA and RalB in in vivo hamster tumor model.Cancer Cell Int. 2011 Jun 29;11(1):22. doi: 10.1186/1475-2867-11-22. Cancer Cell Int. 2011. PMID: 21714887 Free PMC article.
-
Secreted Amyloid Precursor Protein Alpha, a Neuroprotective Protein in the Brain Has Widespread Effects on the Transcriptome and Proteome of Human Inducible Pluripotent Stem Cell-Derived Glutamatergic Neurons Related to Memory Mechanisms.Front Neurosci. 2022 May 26;16:858524. doi: 10.3389/fnins.2022.858524. eCollection 2022. Front Neurosci. 2022. PMID: 35692428 Free PMC article.
References
-
- Ahnert-Hilger G, Wegenhorst U, Stecher B, Spicher K, Rosenthal W, Gratz M. Exocytosis from permeabilized bovine adrenal chromaffin cells is differently modulated by guanosine 5′-[γ-thio]triphosphate and guanosine 5′-[βγ-imido]triphosphate. Evidence for the involvement of various guanine nucleotide-binding proteins. Biochem J. 1992;284:321–326. - PMC - PubMed
-
- Arac D, Chen X, Khant HA, Ubach J, Ludtke SJ, Kikkawa M, Johnson AE, Chiu W, Südhof TC, Rizo J. Close membrane–membrane proximity induced by Ca2+-dependent multivalent binding of synaptotagmin-1 to phospholipids. Nat Struct Mol Biol. 2006;13:209–217. - PubMed
-
- Banerjee A, Kowalchyk JA, DasGupta BR, Martin TF. SNAP-25 is required for a late postdocking step in Ca2+-dependent exocytosis. J Biol Chem. 1996;271:20227–20230. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
