Is HIV-1 evolving to a less virulent form in humans?
- PMID: 17203103
- PMCID: PMC7097722
- DOI: 10.1038/nrmicro1594
Is HIV-1 evolving to a less virulent form in humans?
Abstract
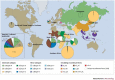
During the rapid spread of HIV-1 in humans, the main (M) group of HIV-1 has evolved into ten distinct subtypes, undergone countless recombination events and diversified extensively. The impact of this extreme genetic diversity on the phenotype of HIV-1 has only recently become a research focus, but early findings indicate that the dominance of HIV-1 subtype C in the current epidemic might be related to the lower virulence of this subtype compared with other subtypes. Here, we explore whether HIV-1 has reached peak virulence or has already started the slow path to attenuation.
Conflict of interest statement
The authors declare no competing financial interests.
Figures



Comment in
-
HIV-1 over time: fitness loss or robustness gain?Nat Rev Microbiol. 2007 Sep;5(9):C1. doi: 10.1038/nrmicro1594-c1. Nat Rev Microbiol. 2007. PMID: 17703224 No abstract available.
References
-
- Lemey P, Pybus OG, Van DS, Vandamme AM. A Bayesian statistical analysis of human T-cell lymphotropic virus evolutionary rates. Infect. Genet. Evol. 2005;5:291–298. - PubMed
-
- Van DS, Salemi M, Vandamme AM. Dating the origin of the African human T-cell lymphotropic virus type-i (HTLV-I) subtypes. Mol. Biol. Evol. 2001;18:661–671. - PubMed
-
- Yoshida M. Discovery of HTLV-1, the first human retrovirus, its unique regulatory mechanisms, and insights into pathogenesis. Oncogene. 2005;24:5931–5937. - PubMed
-
- Korber B, et al. Timing the ancestor of the HIV-1 pandemic strains. Science. 2000;288:1789–1796. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

