An NF-kappaB and slug regulatory loop active in early vertebrate mesoderm
- PMID: 17205110
- PMCID: PMC1762408
- DOI: 10.1371/journal.pone.0000106
An NF-kappaB and slug regulatory loop active in early vertebrate mesoderm
Abstract
Background: In both Drosophila and the mouse, the zinc finger transcription factor Snail is required for mesoderm formation; its vertebrate paralog Slug (Snai2) appears to be required for neural crest formation in the chick and the clawed frog Xenopus laevis. Both Slug and Snail act to induce epithelial to mesenchymal transition (EMT) and to suppress apoptosis.
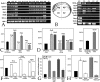
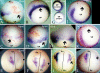
Methodology & principle findings: Morpholino-based loss of function studies indicate that Slug is required for the normal expression of both mesodermal and neural crest markers in X. laevis. Both phenotypes are rescued by injection of RNA encoding the anti-apoptotic protein Bcl-xL; Bcl-xL's effects are dependent upon IkappaB kinase-mediated activation of the bipartite transcription factor NF-kappaB. NF-kappaB, in turn, directly up-regulates levels of Slug and Snail RNAs. Slug indirectly up-regulates levels of RNAs encoding the NF-kappaB subunit proteins RelA, Rel2, and Rel3, and directly down-regulates levels of the pro-apopotic Caspase-9 RNA.
Conclusions/significance: These studies reveal a Slug/Snail-NF-kappaB regulatory circuit, analogous to that present in the early Drosophila embryo, active during mesodermal formation in Xenopus. This is a regulatory interaction of significance both in development and in the course of inflammatory and metastatic disease.
Conflict of interest statement
Figures



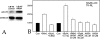
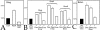
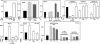
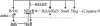
Similar articles
-
Ajuba LIM proteins are snail/slug corepressors required for neural crest development in Xenopus.Dev Cell. 2008 Mar;14(3):424-36. doi: 10.1016/j.devcel.2008.01.005. Dev Cell. 2008. PMID: 18331720 Free PMC article.
-
Snail precedes slug in the genetic cascade required for the specification and migration of the Xenopus neural crest.Development. 2003 Feb;130(3):483-94. doi: 10.1242/dev.00238. Development. 2003. PMID: 12490555
-
YY1 regulates the neural crest-associated slug gene in Xenopus laevis.J Biol Chem. 2004 Nov 5;279(45):46826-34. doi: 10.1074/jbc.M406140200. Epub 2004 Aug 23. J Biol Chem. 2004. PMID: 15326190
-
The activated NF-kappaB-Snail-RKIP circuitry in cancer regulates both the metastatic cascade and resistance to apoptosis by cytotoxic drugs.Crit Rev Immunol. 2009;29(3):241-54. doi: 10.1615/critrevimmunol.v29.i3.40. Crit Rev Immunol. 2009. PMID: 19538137 Review.
-
Snail/slug family of repressors: slowly going into the fast lane of development and cancer.Gene. 2000 Oct 17;257(1):1-12. doi: 10.1016/s0378-1119(00)00371-1. Gene. 2000. PMID: 11054563 Review.
Cited by
-
Identifying domains of EFHC1 involved in ciliary localization, ciliogenesis, and the regulation of Wnt signaling.Dev Biol. 2016 Mar 15;411(2):257-265. doi: 10.1016/j.ydbio.2016.01.004. Epub 2016 Jan 16. Dev Biol. 2016. PMID: 26783883 Free PMC article.
-
Zoledronic acid suppresses metastasis of esophageal squamous cell carcinoma cells through upregulating the tight junction protein occludin.Cytotechnology. 2016 Aug;68(4):1233-41. doi: 10.1007/s10616-015-9884-7. Epub 2015 Jul 24. Cytotechnology. 2016. PMID: 26204820 Free PMC article.
-
Snail2/Slug cooperates with Polycomb repressive complex 2 (PRC2) to regulate neural crest development.Development. 2015 Feb 15;142(4):722-31. doi: 10.1242/dev.111997. Epub 2015 Jan 23. Development. 2015. PMID: 25617436 Free PMC article.
-
Neural crest development in Xenopus requires Protocadherin 7 at the lateral neural crest border.Mech Dev. 2018 Feb;149:41-52. doi: 10.1016/j.mod.2018.01.002. Epub 2018 Jan 31. Mech Dev. 2018. PMID: 29366801 Free PMC article.
-
Chibby functions in Xenopus ciliary assembly, embryonic development, and the regulation of gene expression.Dev Biol. 2014 Nov 15;395(2):287-98. doi: 10.1016/j.ydbio.2014.09.008. Epub 2014 Sep 16. Dev Biol. 2014. PMID: 25220153 Free PMC article.
References
-
- Thiery JP, Sleeman JP. Complex networks orchestrate epithelial-mesenchymal transitions. Nat Rev Mol Cell Biol. 2006;7:131–42. - PubMed
-
- Fritzenwanker JH, Saina M, Technau U. Analysis of forkhead and snail expression reveals epithelial-mesenchymal transitions during embryonic and larval development of Nematostella vectensis. Dev Biol. 2004;275:389–402. - PubMed
-
- Technau U, Scholz CB. Origin and evolution of endoderm and mesoderm. Int J Dev Biol. 2003;47:531–9. - PubMed
-
- Vickaryous MK, Hall BK. Human cell type diversity, evolution, development, and classification with special reference to cells derived from the neural crest. Biol Rev Camb Philos Soc. 2006;81:425–55. - PubMed
-
- Whiteley M, Noguchi PD, Sensabaugh SM, Odenwald WF, Kassis JA. The Drosophila gene escargot encodes a zinc finger motif found in snail-related genes. Mech Dev. 1992;36:117–27. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

