Functional characterization and conformational analysis of the Herpesvirus saimiri Tip-C484 protein
- PMID: 17207813
- PMCID: PMC2262936
- DOI: 10.1016/j.jmb.2006.12.026
Functional characterization and conformational analysis of the Herpesvirus saimiri Tip-C484 protein
Abstract
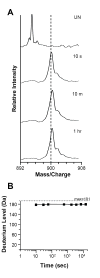
Tyrosine kinase interacting protein (Tip) of Herpesvirus saimiri (HVS) activates the lymphoid-specific member of the Src family kinase Lck. The Tip:Lck interaction is essential for transformation and oncogenesis in HVS-infected cells. As there are no structural data for Tip, hydrogen-exchange mass spectrometry was used to investigate the conformation of a nearly full-length form (residues 1-187) of Tip from HVS strain C484. Disorder predictions suggested that Tip would be mostly unstructured, so great care was taken to ascertain whether recombinant Tip was functional. Circular dichroism and gel-filtration analysis indicated an extended, unstructured protein. In vitro and in vivo binding and kinase assays confirmed that purified, recombinant Tip interacted with Lck, was capable of activating Lck kinase activity strongly and was multiply phosphorylated by Lck. Hydrogen-exchange mass spectrometry of Tip then showed that the majority of backbone amide hydrogen atoms became deuterated after only 10 s of labeling. Such a result suggested that Tip was almost totally unstructured in solution. Digestion of deuterium-labeled Tip revealed some regions with minor protection from exchange. Overall, it was found that, although recombinant Tip is still functional and capable of binding and activating its target Lck, it is largely unstructured.
Figures




Similar articles
-
Characterization of Lck-binding elements in the herpesviral regulatory Tip protein.Biochemistry. 2004 Nov 30;43(47):14932-9. doi: 10.1021/bi0485068. Biochemistry. 2004. PMID: 15554700
-
Growth transformation of human T cells by herpesvirus saimiri requires multiple Tip-Lck interaction motifs.J Virol. 2006 Oct;80(20):9934-42. doi: 10.1128/JVI.01112-06. J Virol. 2006. PMID: 17005671 Free PMC article.
-
The herpesvirus saimiri tip484 and tip488 proteins both stimulate lck tyrosine protein kinase activity in vivo and in vitro.Virology. 2002 Jun 5;297(2):281-8. doi: 10.1006/viro.2002.1419. Virology. 2002. PMID: 12083826
-
Lck protein tyrosine kinase is a key regulator of T-cell activation and a target for signal intervention by Herpesvirus saimiri and other viral gene products.Eur J Biochem. 2000 Jun;267(12):3413-21. doi: 10.1046/j.1432-1327.2000.01412.x. Eur J Biochem. 2000. PMID: 10848956 Review.
-
Mechanisms of cell transformation by Herpesvirus saimiri.Anticancer Res. 1999 Mar-Apr;19(2A):973-83. Anticancer Res. 1999. PMID: 10368641 Review.
Cited by
-
Time window expansion for HDX analysis of an intrinsically disordered protein.J Am Soc Mass Spectrom. 2013 Oct;24(10):1584-92. doi: 10.1007/s13361-013-0669-y. Epub 2013 Jul 25. J Am Soc Mass Spectrom. 2013. PMID: 23884631 Free PMC article.
-
Advances in Hydrogen/Deuterium Exchange Mass Spectrometry and the Pursuit of Challenging Biological Systems.Chem Rev. 2022 Apr 27;122(8):7562-7623. doi: 10.1021/acs.chemrev.1c00279. Epub 2021 Sep 7. Chem Rev. 2022. PMID: 34493042 Free PMC article. Review.
-
Conformational dynamics of the Escherichia coli DNA polymerase manager proteins UmuD and UmuD'.J Mol Biol. 2010 Apr 23;398(1):40-53. doi: 10.1016/j.jmb.2010.02.040. Epub 2010 Mar 4. J Mol Biol. 2010. PMID: 20206636 Free PMC article.
-
Lck-dependent Fyn activation requires C terminus-dependent targeting of kinase-active Lck to lipid rafts.J Biol Chem. 2008 Sep 26;283(39):26409-22. doi: 10.1074/jbc.M710372200. Epub 2008 Jul 27. J Biol Chem. 2008. PMID: 18660530 Free PMC article.
-
The HIV-1 protein Nef activates the Tec family kinase Btk by stabilizing an intermolecular SH3-SH2 domain interaction.Sci Signal. 2022 Sep 20;15(752):eabn8359. doi: 10.1126/scisignal.abn8359. Epub 2022 Sep 20. Sci Signal. 2022. PMID: 36126115 Free PMC article.
References
-
- Brinkmann MM, Schulz TF. Regulation of intracellular signalling by the terminal membrane proteins of members of the Gammaherpesvirinae. J Gen Virol. 2006;87:1047–1074. - PubMed
-
- Biesinger B, Trimble JJ, Desrosiers RC, Fleckenstein B. The divergence between two oncogenic Herpesvirus saimiri strains in a genomic region related to the transforming phenotype. Virology. 1990;176:505–514. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

