Antigen receptor signalling: a distinctive role for the p110delta isoform of PI3K
- PMID: 17208518
- PMCID: PMC2358943
- DOI: 10.1016/j.it.2006.12.007
Antigen receptor signalling: a distinctive role for the p110delta isoform of PI3K
Abstract
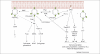
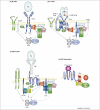
The activation of antigen receptors triggers two important signalling pathways originating from phosphatidylinositol(4,5)-bisphosphate [PtdIns(4,5)P(2)]. The first is phospholipase Cgamma (PLCgamma)-mediated hydrolysis of PtdIns(4,5)P(2), resulting in the activation of Ras, protein kinase C and Ca(2+) flux. This culminates in profound alterations in gene expression and effector-cell responses, including secretory granule exocytosis and cytokine production. By contrast, phosphoinositide 3-kinases (PI3Ks) phosphorylate PtdIns(4,5)P(2) to yield phosphatidylinositol(3,4,5)-trisphosphate, activating signalling pathways that overlap with PLCgamma or are PI3K-specific. Pathways that are PI3K-specific include Akt-mediated inactivation of Foxo transcription factors and transcription-independent regulation of glucose uptake and metabolism. The p110delta isoform of PI3K is the main source of PI3K activity following antigen recognition by B cells, T cells and mast cells. Here, we review the roles of p110delta in regulating antigen-dependent responses in these cell types.
Figures
References
-
- Vanhaesebroeck B. Synthesis and function of 3-phosphorylated inositol lipids. Annu. Rev. Biochem. 2001;70:535–602. - PubMed
-
- Suire S. Gβγs and the Ras binding domain of p110γ are both important regulators of PI3Kγ signalling in neutrophils. Nat. Cell Biol. 2006;8:1303–1309. - PubMed
-
- Ward S.G. T lymphocytes on the move: chemokines, PI 3-kinase and beyond. Trends Immunol. 2006;27:80–87. - PubMed
-
- Sasaki T. Function of PI3Kγ in thymocyte development, T cell activation, and neutrophil migration. Science. 2000;287:1040–1046. - PubMed
-
- Laffargue M. Phosphoinositide 3-kinase γ is an essential amplifier of mast cell function. Immunity. 2002;16:441–451. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

