Pharmacokinetic interaction between darunavir boosted with ritonavir and omeprazole or ranitidine in human immunodeficiency virus-negative healthy volunteers
- PMID: 17210768
- PMCID: PMC1803132
- DOI: 10.1128/AAC.01203-06
Pharmacokinetic interaction between darunavir boosted with ritonavir and omeprazole or ranitidine in human immunodeficiency virus-negative healthy volunteers
Abstract
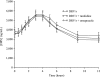
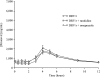
Darunavir (DRV; TMC114; Prezista) is a human immunodeficiency virus (HIV) protease inhibitor used in combination with low-dose ritonavir (RTV) (DRV/r) as a pharmacokinetic enhancer. Protease inhibitor absorption may be decreased during coadministration of drugs that limit stomach acid secretion and increase gastric pH. This study was conducted to investigate the effect of ranitidine and omeprazole on the plasma pharmacokinetics of DRV and RTV in HIV-negative healthy volunteers. Sixteen volunteers completed the study and received DRV/r, DRV/r plus ranitidine, and DRV/r plus omeprazole, in three separate sessions. Treatment was given for 4 days with an additional morning dose on day 5, and regimens were separated by a washout period of 7 days. Samples were taken over a 12-h period on day 5 for the assessment of DRV and RTV plasma concentrations. Pharmacokinetic parameters assessed included DRV area under the curve, maximum plasma concentration, and trough plasma concentration. The least-squares mean ratios and 90% confidence intervals are reported with treatment of DRV/r alone as a reference. Compared with DRV/r alone, no significant changes in DRV pharmacokinetic parameters were observed during coadministration of DRV/r and either ranitidine or omeprazole. Treatment regimens were generally well tolerated, and no serious adverse events were reported. In conclusion, coadministration of DRV/r and ranitidine or omeprazole was well tolerated by the volunteers. Ranitidine and omeprazole did not have a significant influence on DRV pharmacokinetics. No dose adjustments are required when DRV/r is coadministered with omeprazole or ranitidine.
Figures
Similar articles
-
Clinical pharmacokinetics of darunavir.Clin Pharmacokinet. 2007;46(9):739-56. doi: 10.2165/00003088-200746090-00002. Clin Pharmacokinet. 2007. PMID: 17713972 Review.
-
Effects of acid-reducing agents on the pharmacokinetics of lopinavir/ritonavir and ritonavir-boosted atazanavir.J Clin Pharmacol. 2008 May;48(5):553-62. doi: 10.1177/0091270007313392. J Clin Pharmacol. 2008. PMID: 18440920 Clinical Trial.
-
Pharmacokinetic interaction between indinavir and darunavir with low-dose ritonavir in healthy volunteers.Intervirology. 2010;53(3):176-82. doi: 10.1159/000289341. Epub 2010 Mar 3. Intervirology. 2010. PMID: 20197684
-
Pharmacokinetic interaction between darunavir and saquinavir in HIV-negative volunteers.Ther Drug Monit. 2007 Dec;29(6):795-801. doi: 10.1097/FTD.0b013e31815d23e6. Ther Drug Monit. 2007. PMID: 18043478 Clinical Trial.
-
Darunavir: pharmacokinetics and drug interactions.Antivir Ther. 2008;13(1):1-13. Antivir Ther. 2008. PMID: 18389894 Review.
Cited by
-
Darunavir: in the treatment of HIV-1 infection.Drugs. 2007;67(18):2791-801. doi: 10.2165/00003495-200767180-00010. Drugs. 2007. PMID: 18062724 Review.
-
Application of Physiologically-Based Pharmacokinetic Modeling to Predict Gastric pH-Dependent Drug-Drug Interactions for Weak Base Drugs.CPT Pharmacometrics Syst Pharmacol. 2020 Aug;9(8):456-465. doi: 10.1002/psp4.12541. Epub 2020 Jul 31. CPT Pharmacometrics Syst Pharmacol. 2020. PMID: 32633893 Free PMC article.
-
Clinical pharmacokinetics of darunavir.Clin Pharmacokinet. 2007;46(9):739-56. doi: 10.2165/00003088-200746090-00002. Clin Pharmacokinet. 2007. PMID: 17713972 Review.
-
Inflammation Induces Changes in the Functional Expression of P-gp, BCRP, and MRP2: An Overview of Different Models and Consequences for Drug Disposition.Pharmaceutics. 2021 Sep 23;13(10):1544. doi: 10.3390/pharmaceutics13101544. Pharmaceutics. 2021. PMID: 34683838 Free PMC article. Review.
-
A pharmacokinetic study of etravirine (TMC125) co-administered with ranitidine and omeprazole in HIV-negative volunteers.Br J Clin Pharmacol. 2008 Oct;66(4):508-16. doi: 10.1111/j.1365-2125.2008.03214.x. Epub 2008 Apr 25. Br J Clin Pharmacol. 2008. PMID: 18492125 Free PMC article. Clinical Trial.
References
-
- Agarwala, S., T. Eley, M. Child, Y. Wang, E. Hughes, E. Chung, R. Wang, and D. Grasela. 2005. Pharmacokinetic effect of famotidine on atazanavir with and without ritonavir in healthy subjects, abstr. 11. Sixth Int. Workshop Clin. Pharmacol. HIV Ther., Montreal, Quebec, Canada, 28 to 30 April 2005.
-
- Agarwala, S., K. Gray, Y. Wang, and D. Grasela. 2005. Pharmacokinetic effect of omeprazole on atazanavir co-administered with ritonavir in healthy subjects, abstr. 658. Twelfth Conf. Retrovir. Opportun. Infect., Boston, MA, 22 to 25 February 2005.
-
- Bouche, M. P., L. Michielsen, M. Piot, and P. Timmerman. 2006. Swift and simultaneous determination of darunavir (TMC114) and ritonavir in human plasma using LC-MS/MS, abstr. TuP-042. Seventeenth Int. Mass Spectrom. Conf., Prague, Czech Republic, 27 August to 1 September 2006.
-
- Bristol-Myers Squibb. 2004. Reyataz (atazanavir). Prescribing information. Bristol-Myers Squibb, Princeton, NJ.
-
- De Meyer, S., H. Azijn, D. Surleraux, D. Jochmans, A. Tahri, R. Pauwels, P. Wigerinck, and M. P. de Bethune. 2005. TMC114, a novel human immunodeficiency virus type 1 protease inhibitor active against protease inhibitor-resistant viruses, including a broad range of clinical isolates. Antimicrob. Agents Chemother. 49:2314-2321. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

