XANES measurements of the rate of radiation damage to selenomethionine side chains
- PMID: 17211072
- PMCID: PMC2806430
- DOI: 10.1107/S0909049506048898
XANES measurements of the rate of radiation damage to selenomethionine side chains
Abstract
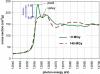
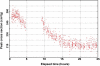
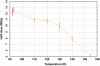
The radiation-induced disordering of selenomethionine (SeMet) side chains represents a significant impediment to protein structure solution. Not only does the increased B-factor of these sites result in a serious drop in phasing power, but some sites decay much faster than others in the same unit cell. These radio-labile SeMet side chains decay faster than high-order diffraction spots with dose, making it difficult to detect this kind of damage by inspection of the diffraction pattern. The selenium X-ray absorbance near-edge spectrum (XANES) from samples containing SeMet was found to change significantly after application of X-ray doses of 10-100 MGy. Most notably, the sharp ;white line' feature near the canonical Se edge disappears. The change was attributed to breakage of the Cgamma-Se bond in SeMet. This spectral change was used as a probe to measure the decay rate of SeMet with X-ray dose in cryo-cooled samples. Two protein crystal types and 15 solutions containing free SeMet amino acid were examined. The damage rate was influenced by the chemical and physical condition of the sample, and the half-decaying dose for the selenium XANES signal ranged from 5 to 43 MGy. These decay rates were 34- to 3.8-fold higher than the rate at which the Se atoms interacted directly with X-ray photons, so the damage mechanism must be a secondary effect. Samples that cooled to a more crystalline state generally decayed faster than samples that cooled to an amorphous solid. The single exception was a protein crystal where a nanocrystalline cryoprotectant had a protective effect. Lowering the pH, especially with ascorbic or nitric acids, had a protective effect, and SeMet lifetime increased monotonically with decreasing sample temperature (down to 93 K). The SeMet lifetime in one protein crystal was the same as that of the free amino acid, and the longest SeMet lifetime measured was found in the other protein crystal type. This protection was found to arise from the folded structure of the protein molecule. A mechanism to explain observed decay rates involving the damaging species following the electric field lines around protein molecules is proposed.
Figures



Similar articles
-
The many faces of radiation-induced changes.J Synchrotron Radiat. 2007 Jan;14(Pt 1):24-33. doi: 10.1107/S0909049506046589. Epub 2006 Dec 15. J Synchrotron Radiat. 2007. PMID: 17211069
-
Modelling and refining site-specific radiation damage in SAD/MAD phasing.J Synchrotron Radiat. 2007 Jan;14(Pt 1):34-42. doi: 10.1107/S0909049506038970. Epub 2006 Dec 15. J Synchrotron Radiat. 2007. PMID: 17211070
-
A comparison of SAD and two-wavelength MAD phasing for radiation-damaged Se-MET crystals.J Synchrotron Radiat. 2007 Jan;14(Pt 1):43-50. doi: 10.1107/S0909049506041045. Epub 2006 Dec 15. J Synchrotron Radiat. 2007. PMID: 17211071
-
Radiation damage to protein specimens from electron beam imaging and diffraction: a mini-review of anti-damage approaches, with special reference to synchrotron X-ray crystallography.J Synchrotron Radiat. 2007 Jan;14(Pt 1):116-27. doi: 10.1107/S0909049506052307. Epub 2006 Dec 15. J Synchrotron Radiat. 2007. PMID: 17211078 Review.
-
Radiation damage in protein crystals examined under various conditions by different methods.J Synchrotron Radiat. 2009 Mar;16(Pt 2):129-32. doi: 10.1107/S0909049509005238. Epub 2009 Feb 25. J Synchrotron Radiat. 2009. PMID: 19240324 Review.
Cited by
-
To scavenge or not to scavenge, that is STILL the question.J Synchrotron Radiat. 2013 Jan;20(Pt 1):23-36. doi: 10.1107/S0909049512046237. Epub 2012 Dec 5. J Synchrotron Radiat. 2013. PMID: 23254653 Free PMC article.
-
RNA protects a nucleoprotein complex against radiation damage.Acta Crystallogr D Struct Biol. 2016 May;72(Pt 5):648-57. doi: 10.1107/S2059798316003351. Epub 2016 Apr 26. Acta Crystallogr D Struct Biol. 2016. PMID: 27139628 Free PMC article.
-
Using XAS to monitor radiation damage in real time and post-analysis, and investigation of systematic errors of fluorescence XAS for Cu-bound amyloid-β.J Appl Crystallogr. 2024 Feb 1;57(Pt 1):125-139. doi: 10.1107/S1600576723010890. eCollection 2024 Feb 1. J Appl Crystallogr. 2024. PMID: 38322727 Free PMC article.
-
Identification of patterns in diffraction intensities affected by radiation exposure.J Synchrotron Radiat. 2013 Jan;20(Pt 1):37-48. doi: 10.1107/S0909049512048807. Epub 2012 Dec 6. J Synchrotron Radiat. 2013. PMID: 23254654 Free PMC article.
-
The design of macromolecular crystallography diffraction experiments.Acta Crystallogr D Biol Crystallogr. 2011 Apr;67(Pt 4):261-70. doi: 10.1107/S0907444911007608. Epub 2011 Mar 18. Acta Crystallogr D Biol Crystallogr. 2011. PMID: 21460444 Free PMC article.
References
-
- Banumathi S, Zwart PH, Ramagopal UA, Dauter M, Dauter Z. Acta Cryst. 2004;D60:1085–1093. - PubMed
-
- Box HR. Radiation Effects: ESR and ENDOR Analysis. Academic Press; London: 1977.
-
- Bricogne G, Capelli SC, Evans G, Mitschler A, Pattison P, Roversi P, Schiltz M. J. Appl. Cryst. 2005;38:168–182.
-
- Burmeister WP. Acta Cryst. 2000;D56:328–341. - PubMed
-
- Carugo O, Carugo KD. Trends Biochem. Sci. 2005;30:213–219. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

