Enhanced T-cell reconstitution by hematopoietic progenitors expanded ex vivo using the Notch ligand Delta1
- PMID: 17213287
- PMCID: PMC1852253
- DOI: 10.1182/blood-2006-08-039842
Enhanced T-cell reconstitution by hematopoietic progenitors expanded ex vivo using the Notch ligand Delta1
Abstract
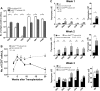
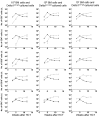
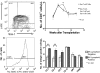
A physiologic role for Notch signaling in hematopoiesis has been clearly defined in lymphoid differentiation, with evidence suggesting a critical role in T-cell versus B-cell fate decisions. Previously, we demonstrated that activation of endogenous Notch receptors by culture of murine lin(-)Sca-1(+)c-kit(+) (LSK) hematopoietic progenitors with exogenously presented Notch ligand, Delta1(ext-IgG), consisting of the extracellular domain of Delta1 fused to the Fc domain of human IgG(1), promoted early T-cell differentiation and increased the number of progenitors capable of short-term lymphoid and myeloid reconstitution. Here we show that culture of LSK precursors with Delta1(ext-IgG) increases the number of progenitors that are able to rapidly repopulate the thymus and accelerate early T-cell reconstitution with a diversified T-cell receptor repertoire. Most of the early T-cell reconstitution originated from cells that expressed lymphoid-associated antigens: B220, Thy1, CD25, and/or IL7Ralpha, whereas the most efficient thymic repopulation on a per cell basis originated from the smaller number of cultured cells that did not express lymphoid-associated antigens. These findings demonstrate the potential of Delta1(ext-IgG)-cultured cells for accelerating early immune reconstitution after hematopoietic cell transplantation.
Figures

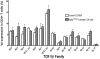

Similar articles
-
Density of the Notch ligand Delta1 determines generation of B and T cell precursors from hematopoietic stem cells.J Exp Med. 2005 May 2;201(9):1361-6. doi: 10.1084/jem.20042450. Epub 2005 Apr 25. J Exp Med. 2005. PMID: 15851488 Free PMC article.
-
Combined effects of Notch signaling and cytokines induce a multiple log increase in precursors with lymphoid and myeloid reconstituting ability.Blood. 2003 Mar 1;101(5):1784-9. doi: 10.1182/blood-2002-06-1862. Epub 2002 Oct 31. Blood. 2003. PMID: 12411302
-
Notch increases T/NK potential of human hematopoietic progenitors and inhibits B cell differentiation at a pro-B stage.Stem Cells. 2009 Jul;27(7):1676-85. doi: 10.1002/stem.94. Stem Cells. 2009. PMID: 19544442
-
[Human hematopoiesis: from CD34 cells to T lymphocytes].Med Sci (Paris). 2007 Feb;23(2):151-9. doi: 10.1051/medsci/2007232151. Med Sci (Paris). 2007. PMID: 17291424 Review. French.
-
Notch signaling in hematopoietic cell transplantation and T cell alloimmunity.Blood Rev. 2013 Nov;27(6):269-77. doi: 10.1016/j.blre.2013.08.001. Epub 2013 Aug 31. Blood Rev. 2013. PMID: 24050990 Free PMC article. Review.
Cited by
-
T cell regeneration after immunological injury.Nat Rev Immunol. 2021 May;21(5):277-291. doi: 10.1038/s41577-020-00457-z. Epub 2020 Oct 23. Nat Rev Immunol. 2021. PMID: 33097917 Free PMC article. Review.
-
Proteins and small molecules for cellular regenerative medicine.Physiol Rev. 2013 Jan;93(1):311-25. doi: 10.1152/physrev.00005.2012. Physiol Rev. 2013. PMID: 23303911 Free PMC article. Review.
-
Producing proT cells to promote immunotherapies.Int Immunol. 2018 Nov 14;30(12):541-550. doi: 10.1093/intimm/dxy051. Int Immunol. 2018. PMID: 30102361 Free PMC article. Review.
-
Advances in umbilical cord blood manipulation-from niche to bedside.Nat Rev Clin Oncol. 2015 Mar;12(3):163-74. doi: 10.1038/nrclinonc.2014.215. Epub 2014 Dec 16. Nat Rev Clin Oncol. 2015. PMID: 25511187 Free PMC article. Review.
-
A robust approach for the generation of functional hematopoietic progenitor cell lines to model leukemic transformation.Blood Adv. 2021 Jan 12;5(1):39-53. doi: 10.1182/bloodadvances.2020003022. Blood Adv. 2021. PMID: 33570624 Free PMC article.
References
-
- Ochs L, Shu XO, Miller J, et al. Late infections after allogeneic bone marrow transplantations: comparison of incidence in related and unrelated donor transplant recipients. Blood. 1995;86:3979–3986. - PubMed
-
- Mackall CL, Bare CV, Granger LA, Sharrow SO, Titus JA, Gress RE. Thymic-independent T cell regeneration occurs via antigen-driven expansion of peripheral T cells resulting in a repertoire that is limited in diversity and prone to skewing. J Immunol. 1996;156:4609–4616. - PubMed
-
- Mackall CL, Gress RE. Pathways of T-cell regeneration in mice and humans: implications for bone marrow transplantation and immunotherapy. Immunol Rev. 1997;157:61–72. - PubMed
-
- Gorski J, Yassai M, Zhu X, et al. Circulating T cell repertoire complexity in normal individuals and bone marrow recipients analyzed by CDR3 size spectratyping: correlation with immune status. J Immunol. 1994;152:5109–5119. - PubMed
-
- Talvensaari K, Clave E, Douay C, et al. A broad T-cell repertoire diversity and an efficient thymic function indicate a favorable long-term immune reconstitution after cord blood stem cell transplantation. Blood. 2002;99:1458–1464. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

