Negative feedback loop in T cell activation through IkappaB kinase-induced phosphorylation and degradation of Bcl10
- PMID: 17213322
- PMCID: PMC1783413
- DOI: 10.1073/pnas.0606982104
Negative feedback loop in T cell activation through IkappaB kinase-induced phosphorylation and degradation of Bcl10
Abstract
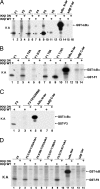
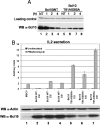
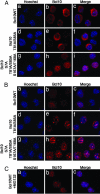
Activation of the transcription factor NF-kappaB after stimulation through antigen receptors is important for lymphocyte differentiation, activation, proliferation, and protection against apoptosis. Much progress has been made in understanding the molecular events leading to NF-kappaB activation, but how this activation is eventually down-regulated is less well understood. Recent studies have indicated that Bcl10 functions downstream of lymphocyte antigen receptors to promote the activation of the IkappaB kinase complex leading to the phosphorylation and degradation of the IkappaB inhibitors of NF-kappaB. Bcl10 has also been implicated in the pathogenesis of mucosa-associated lymphoid tissue lymphoma, possibly in association with its nuclear localization. Here, we provide evidence that the IkappaB kinase complex phosphorylates Bcl10 after T cell antigen receptor stimulation and causes its proteolysis via the beta-TrCP ubiquitin ligase/proteasome pathway. These findings document a negative regulatory activity of the IKK complex and suggest that Bcl10 degradation is part of the regulatory mechanisms that precisely control the response to antigens. Mutants of Bcl10 in the IKK phosphorylation site are resistant to degradation, accumulate in the nucleus, and lead to an increase in IL-2 production after T cell antigen receptor stimulation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
CD28 stimulation triggers NF-kappaB activation through the CARMA1-PKCtheta-Grb2/Gads axis.Int Immunol. 2008 Dec;20(12):1507-15. doi: 10.1093/intimm/dxn108. Epub 2008 Oct 1. Int Immunol. 2008. PMID: 18829987
-
Striking back at the activator: how IkappaB kinase terminates antigen receptor responses.Sci STKE. 2007 May 1;2007(384):pe19. doi: 10.1126/stke.3842007pe19. Sci STKE. 2007. PMID: 17473308
-
Post-translational modifications regulate distinct functions of CARMA1 and BCL10.Trends Immunol. 2007 Jun;28(6):281-8. doi: 10.1016/j.it.2007.04.004. Epub 2007 Apr 30. Trends Immunol. 2007. PMID: 17468049 Review.
-
CaMKII targets Bcl10 in T-cell receptor induced activation of NF-κB.Mol Immunol. 2011 Jul;48(12-13):1448-60. doi: 10.1016/j.molimm.2011.03.020. Epub 2011 Apr 22. Mol Immunol. 2011. PMID: 21513986
-
NF-kappaB signaling in lymphocytes: a new cast of characters.J Cell Sci. 2004 Jan 1;117(Pt 1):31-9. doi: 10.1242/jcs.00904. J Cell Sci. 2004. PMID: 14657271 Review.
Cited by
-
Regulation of T cell function by the ubiquitin-specific protease USP9X via modulating the Carma1-Bcl10-Malt1 complex.Proc Natl Acad Sci U S A. 2013 Jun 4;110(23):9433-8. doi: 10.1073/pnas.1221925110. Epub 2013 May 20. Proc Natl Acad Sci U S A. 2013. PMID: 23690623 Free PMC article.
-
The Global Phosphorylation Landscape of SARS-CoV-2 Infection.Cell. 2020 Aug 6;182(3):685-712.e19. doi: 10.1016/j.cell.2020.06.034. Epub 2020 Jun 28. Cell. 2020. PMID: 32645325 Free PMC article.
-
Virgin olive oil rich in phenolic compounds modulates the expression of atherosclerosis-related genes in vascular endothelium.Eur J Nutr. 2016 Mar;55(2):519-527. doi: 10.1007/s00394-015-0868-3. Epub 2015 Mar 4. Eur J Nutr. 2016. PMID: 25733165
-
A quantitative signaling screen identifies CARD11 mutations in the CARD and LATCH domains that induce Bcl10 ubiquitination and human lymphoma cell survival.Mol Cell Biol. 2013 Jan;33(2):429-43. doi: 10.1128/MCB.00850-12. Epub 2012 Nov 12. Mol Cell Biol. 2013. PMID: 23149938 Free PMC article.
-
Mechanisms of Regulated and Dysregulated CARD11 Signaling in Adaptive Immunity and Disease.Front Immunol. 2018 Sep 19;9:2105. doi: 10.3389/fimmu.2018.02105. eCollection 2018. Front Immunol. 2018. PMID: 30283447 Free PMC article. Review.
References
-
- Weil R, Israël A. Cell Death Differ. 2006;13:826–833. - PubMed
-
- Bertin J, Wang L, Guo Y, Jacobson MD, Poyet JL, Srinivasula SM, Merriam S, DiStefano PS, Alnemri ES. J Biol Chem. 2001;276:11877–11882. - PubMed
-
- Gaide O, Martinon F, Micheau O, Bonnet D, Thome M, Tschopp J. FEBS Lett. 2001;496:121–127. - PubMed
-
- McAllister-Lucas LM, Inohara N, Lucas PC, Ruland J, Benito A, Li Q, Chen S, Chen FF, Yamaoka S, Verma IM, et al. J Biol Chem. 2001;276:30589–30597. - PubMed
-
- Wang L, Guo Y, Huang WJ, Ke X, Poyet JL, Manji GA, Merriam S, Glucksmann MA, DiStefano PS, Alnemri ES, Bertin J. J Biol Chem. 2001;276:21405–21409. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

