Electron tomography of nascent herpes simplex virus virions
- PMID: 17215293
- PMCID: PMC1865967
- DOI: 10.1128/JVI.02571-06
Electron tomography of nascent herpes simplex virus virions
Abstract
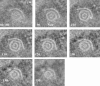
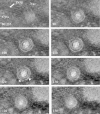
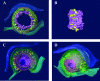
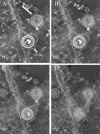
Cells infected with herpes simplex virus type 1 (HSV-1) were conventionally embedded or freeze substituted after high-pressure freezing and stained with uranyl acetate. Electron tomograms of capsids attached to or undergoing envelopment at the inner nuclear membrane (INM), capsids within cytoplasmic vesicles near the nuclear membrane, and extracellular virions revealed the following phenomena. (i) Nucleocapsids undergoing envelopment at the INM, or B capsids abutting the INM, were connected to thickened patches of the INM by fibers 8 to 19 nm in length and < or =5 nm in width. The fibers contacted both fivefold symmetrical vertices (pentons) and sixfold symmetrical faces (hexons) of the nucleocapsid, although relative to the respective frequencies of these subunits in the capsid, fibers engaged pentons more frequently than hexons. (ii) Fibers of similar dimensions bridged the virion envelope and surface of the nucleocapsid in perinuclear virions. (iii) The tegument of perinuclear virions was considerably less dense than that of extracellular virions; connecting fibers were observed in the former case but not in the latter. (iv) The prominent external spikes emanating from the envelope of extracellular virions were absent from perinuclear virions. (v) The virion envelope of perinuclear virions appeared denser and thicker than that of extracellular virions. (vi) Vesicles near, but apparently distinct from, the nuclear membrane in single sections were derived from extensions of the perinuclear space as seen in the electron tomograms. These observations suggest very different mechanisms of tegumentation and envelopment in extracellular compared with perinuclear virions and are consistent with application of the final tegument to unenveloped nucleocapsids in a compartment(s) distinct from the perinuclear space.
Figures


References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

