In vivo imaging of molecularly targeted phage
- PMID: 17217618
- PMCID: PMC1783712
- DOI: 10.1593/neo.06610
In vivo imaging of molecularly targeted phage
Abstract
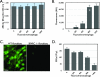
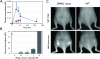
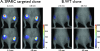
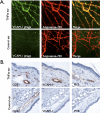
Rapid identification of in vivo affinity ligands would have far-reaching applications for imaging specific molecular targets, in vivo systems imaging, and medical use. We have developed a high-throughput method for identifying and optimizing ligands to map and image biologic targets of interest in vivo. We directly labeled viable phage clones with far-red fluorochromes and comparatively imaged them in vivo by multichannel fluorescence ratio imaging. Using Secreted Protein Acidic and Rich in Cysteine (osteonectin) and vascular cell adhesion molecule-1 as model targets, we show that: 1) fluorescently labeled phage retains target specificity on labeling; 2) in vivo distribution can be quantitated (detection thresholds of approximately 300 phage/mm(3) tissue) throughout the entire depth of the tumor using fluorescent tomographic imaging; and 3) fluorescently labeled phage itself can serve as a replenishable molecular imaging agent. The described method should find widespread application in the rapid in vivo discovery and validation of affinity ligands and, importantly, in the use of fluorochrome-labeled phage clones as in vivo imaging agents.
Figures


Similar articles
-
In vivo selection of phage for the optical imaging of PC-3 human prostate carcinoma in mice.Neoplasia. 2006 Sep;8(9):772-80. doi: 10.1593/neo.06331. Neoplasia. 2006. PMID: 16984734 Free PMC article.
-
In vitro high throughput phage display selection of ovarian cancer avid phage clones for near-infrared optical imaging.Comb Chem High Throughput Screen. 2014;17(10):859-67. doi: 10.2174/1386207317666141031152828. Comb Chem High Throughput Screen. 2014. PMID: 25360673
-
Direct fluorochrome labeling of phage display library clones for studying binding specificities: applications in flow cytometry and fluorescence microscopy.J Immunol Methods. 2004 Dec;295(1-2):119-27. doi: 10.1016/j.jim.2004.09.011. Epub 2004 Oct 27. J Immunol Methods. 2004. PMID: 15627617
-
In vivo phage display--a discovery tool in molecular biomedicine.Biotechnol Adv. 2013 Dec;31(8):1247-59. doi: 10.1016/j.biotechadv.2013.04.004. Epub 2013 Apr 23. Biotechnol Adv. 2013. PMID: 23623852 Review.
-
Advances in phage display technology for drug discovery.Expert Opin Drug Discov. 2015 Jun;10(6):651-69. doi: 10.1517/17460441.2015.1037738. Epub 2015 Apr 24. Expert Opin Drug Discov. 2015. PMID: 25910798 Review.
Cited by
-
Phage-like particle vaccines are highly immunogenic and protect against pathogenic coronavirus infection and disease.NPJ Vaccines. 2022 May 26;7(1):57. doi: 10.1038/s41541-022-00481-1. NPJ Vaccines. 2022. PMID: 35618725 Free PMC article.
-
Hepatocellular carcinoma-targeted nanoparticles for cancer therapy.Int J Oncol. 2018 Feb;52(2):389-401. doi: 10.3892/ijo.2017.4205. Epub 2017 Nov 16. Int J Oncol. 2018. PMID: 29207071 Free PMC article.
-
Chemically Modifying Viruses for Diverse Applications.ACS Chem Biol. 2016 May 20;11(5):1167-79. doi: 10.1021/acschembio.6b00060. Epub 2016 Mar 21. ACS Chem Biol. 2016. PMID: 26930417 Free PMC article. Review.
-
Phage display peptide probes for imaging early response to bevacizumab treatment.Amino Acids. 2011 Nov;41(5):1103-12. doi: 10.1007/s00726-010-0548-9. Epub 2010 Mar 16. Amino Acids. 2011. PMID: 20232090 Free PMC article.
-
M13 bacteriophage display framework that allows sortase-mediated modification of surface-accessible phage proteins.Bioconjug Chem. 2012 Jul 18;23(7):1478-87. doi: 10.1021/bc300130z. Epub 2012 Jul 3. Bioconjug Chem. 2012. PMID: 22759232 Free PMC article.
References
-
- Hoffman JA, Giraudo E, Singh M, Zhang L, Inoue M, Porkka K, Hanahan D, Ruoslahti E. Progressive vascular changes in a transgenic mouse model of squamous cell carcinoma. Cancer Cell. 2003;4:383–391. - PubMed
-
- Joyce JA, Laakkonen P, Bernasconi M, Bergers G, Ruoslahti E, Hanahan D. Stage-specific vascular markers revealed by phage display in a mouse model of pancreatic islet tumorigenesis. Cancer Cell. 2003;4:393–403. - PubMed
-
- Zurita AJ, Arap W, Pasqualini R. Mapping tumor vascular diversity by screening phage display libraries. J Control Release. 2003;91:183–186. - PubMed
-
- Ladner RC, Sato AK, Gorzelany J, de Souza M. Phage display-derived peptides as therapeutic alternatives to antibodies. Drug Discov Today. 2004;9:525–529. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
