SAG/ROC-SCF beta-TrCP E3 ubiquitin ligase promotes pro-caspase-3 degradation as a mechanism of apoptosis protection
- PMID: 17217622
- PMCID: PMC1783718
- DOI: 10.1593/neo.06568
SAG/ROC-SCF beta-TrCP E3 ubiquitin ligase promotes pro-caspase-3 degradation as a mechanism of apoptosis protection
Abstract
Skp1-cullin-F-box protein (SCF) is a multicomponent E3 ubiquitin (Ub) ligase that ubiquitinates a number of important biologic molecules such as p27, beta-catenin, and IkappaB for proteasomal degradation, thus regulating cell proliferation and survival. One SCF component, SAG/ROC2/Rbx2/Hrt2, a RING finger protein, was first identified as a redox-inducible protein, which, when overexpressed, inhibited apoptosis both in vitro and in vivo. We report here that sensitive to apoptosis gene (SAG), as well as its family member ROC1/Rbx1, bound to the proinactive form of caspase-3 (pro-caspase-3). Binding was likely mediated through F-box protein, beta-transducin repeat-containing protein (beta-TrCP), which binds to the first 38 amino acids of pro-caspase-3. Importantly, beta-TrCP1 expression significantly shortened the protein half-life of pro-caspase-3, whereas expression of a dominant-negative beta-TrCP1 mutant with the F-box domain deleted extended it. An in vitro ubiquitination assay showed that SAG/ROC-SCF(beta-TrCP) promoted ubiquitination of pro-caspase-3. Furthermore, endogenous levels of pro-caspase-3 were decreased by overexpression of SAG/ROC-SCF(beta-TrCP) E3 Ub ligases, but increased on siRNA silencing of SAG, regulator of cullin-1 (ROC1), or beta-TrCPs, leading to increased apoptosis by etoposide and TNF-related apoptosis-inducing ligand through increased activation of caspase-3. Thus, pro-caspase-3 appears to be a substrate of SAG/ROC-SCF(beta-TrCP) E3 Ub ligase, which protects cells from apoptosis through increased apoptosis threshold by reducing the basal level of pro-caspase-3.
Figures



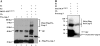

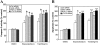
Similar articles
-
SAG/ROC2/Rbx2/Hrt2, a component of SCF E3 ubiquitin ligase: genomic structure, a splicing variant, and two family pseudogenes.DNA Cell Biol. 2001 Jul;20(7):425-34. doi: 10.1089/104454901750361488. DNA Cell Biol. 2001. PMID: 11506706
-
SAG/ROC2/Rbx2 is a novel activator protein-1 target that promotes c-Jun degradation and inhibits 12-O-tetradecanoylphorbol-13-acetate-induced neoplastic transformation.Cancer Res. 2007 Apr 15;67(8):3616-25. doi: 10.1158/0008-5472.CAN-06-4020. Cancer Res. 2007. PMID: 17440073
-
SAG/ROC2/RBX2 is a HIF-1 target gene that promotes HIF-1 alpha ubiquitination and degradation.Oncogene. 2008 Feb 28;27(10):1404-11. doi: 10.1038/sj.onc.1210780. Epub 2007 Sep 10. Oncogene. 2008. PMID: 17828303
-
SAG/ROC/Rbx/Hrt, a zinc RING finger gene family: molecular cloning, biochemical properties, and biological functions.Antioxid Redox Signal. 2001 Aug;3(4):635-50. doi: 10.1089/15230860152542989. Antioxid Redox Signal. 2001. PMID: 11554450 Review.
-
Functional characterization of SAG/RBX2/ROC2/RNF7, an antioxidant protein and an E3 ubiquitin ligase.Protein Cell. 2013 Feb;4(2):103-16. doi: 10.1007/s13238-012-2105-7. Epub 2012 Nov 8. Protein Cell. 2013. PMID: 23136067 Free PMC article. Review.
Cited by
-
Molecular mechanisms of CAND2 in regulating SCF ubiquitin ligases.Nat Commun. 2025 Feb 26;16(1):1998. doi: 10.1038/s41467-025-57065-5. Nat Commun. 2025. PMID: 40011427 Free PMC article.
-
The combination of the prodrugs perforin-CEBPD and perforin-granzyme B efficiently enhances the activation of caspase signaling and kills prostate cancer.Cell Death Dis. 2014 May 8;5(5):e1220. doi: 10.1038/cddis.2014.106. Cell Death Dis. 2014. PMID: 24810056 Free PMC article.
-
The different roles of selective autophagic protein degradation in mammalian cells.Oncotarget. 2015 Nov 10;6(35):37098-116. doi: 10.18632/oncotarget.5776. Oncotarget. 2015. PMID: 26415220 Free PMC article. Review.
-
Functional significance and therapeutic implication of ring-type E3 ligases in colorectal cancer.Oncogene. 2018 Jan 11;37(2):148-159. doi: 10.1038/onc.2017.313. Epub 2017 Sep 18. Oncogene. 2018. PMID: 28925398 Free PMC article. Review.
-
How does caspases regulation play role in cell decisions? apoptosis and beyond.Mol Cell Biochem. 2024 Jul;479(7):1599-1613. doi: 10.1007/s11010-023-04870-5. Epub 2023 Nov 17. Mol Cell Biochem. 2024. PMID: 37976000 Review.
References
-
- Thompson CB. Apoptosis in the pathogenesis and treatment of disease. Science. 1995;267(5203):1456–1462. - PubMed
-
- Wyllie AH, Kerr JF, Currie AR. Cell death: the significance of apoptosis. Int Rev Cytol. 1980;68:251–306. - PubMed
-
- Hengartner MO. The biochemistry of apoptosis. Nature. 2000;407(6805):770–776. - PubMed
-
- Earnshaw WC, Martins LM, Kaufmann SH. Mammalian caspases: structure, activation, substrates, and functions during apoptosis. Annu Rev Biochem. 1999;68:383–424. - PubMed
-
- Cryns V, Yuan J. Proteases to die for. Genes Dev. 1998;12(11):1551–1570. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
