Lymphatic vessels assessment in feline mammary tumours
- PMID: 17222331
- PMCID: PMC1783859
- DOI: 10.1186/1471-2407-7-7
Lymphatic vessels assessment in feline mammary tumours
Abstract
Background: The lymphatic vessels play a crucial role in a variety of human cancers since tumour cell lymphatic invasion significantly influences prognosis. It is not known if pre-existing lymphatics are enough for tumour dissemination or de novo development is necessary. VEGFR-3 is an angiogenetic mediator for both lymphatic and blood vessels during embryonic development, and only for lymphatics after birth. VEGF is a mediator of both vasculogenesis and angiogenesis, regulates the growth of lymphatics in various experimental models, and is produced in many solid tumours. CD44 mediates hyaluronic acid (HA)-dependent cell adhesion: besides promoting invasion, this interaction also supports neoangiogenesis that indirectly stimulates tumour cell proliferation. The expression of VEGF-C (Vascular Endothelial Growth Factor-C), its receptor VEGFR-3 and CD44, were studied on feline mammary samples to assess the importance of lymphangiogenesis and lymphangiotrophism in neoplasia.
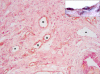
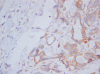
Methods: Samples were taken from six normal mammary glands (NMG), ten benign (BT) and 32 malignant (MT) tumours. Immunohistochemical laminin/VEGFR-3 double stain, VEGF-C and CD44 stains were applied to 4 mum-thick sections, and their expression evaluated in intratumoral/extratumoral and intramammary/extramammary fields.
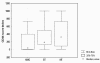
Results: All groups revealed a higher number of lymphatics in the extratumoral/extramammary areas. VEGF-C expression in the epithelium paralleled the number of positive vessels in the NMG, BT and MT, whereas VEGF-C higher expression was noted in the intratumoral fields only in infiltrating MT. CD44 score was lower in extratumoral than intratumoral fields in tumours and showed a significant increase in extramammary/extratumoral fields from NMG to MT. Pearson test showed a significant and inversely proportional correlation between CD44 expression and the number of lymphatic vessels with VEGFR-3 in malignant infiltrating tumours.
Conclusion: The number of both VEGFR-3 positive and negative lymphatics in the extratumoral and extramammary stroma was significantly higher than intratumoral and intramammary fields respectively in the NMG, BT and MT. This suggests a scant biological importance of intratumoral lymphatics while their higher number is due to the concentration of existing vessels following compression of the extratumoral stroma in spite of a non demonstrable increase from NMG to MT. The tumour model employed provided no evidence of lymphangiogenesis, and metastasis in the regional lymph node develops following the spread through the pre-existing lymphatic network.
Figures




Similar articles
-
Immunohistochemical analysis of VEGF-C/VEGFR-3 system and lymphatic vessel extent in normal and adenomatous human pituitary tissues.Exp Clin Endocrinol Diabetes. 2008 Mar;116(3):152-7. doi: 10.1055/s-2007-992120. Epub 2008 Jan 18. Exp Clin Endocrinol Diabetes. 2008. PMID: 18205092
-
Differential expression of VEGF ligands and receptors in prostate cancer.Prostate. 2013 May;73(6):563-72. doi: 10.1002/pros.22596. Epub 2012 Oct 4. Prostate. 2013. PMID: 23038639
-
Lymphangiogenesis and its relationship with lymphatic metastasis and prognosis in malignant melanoma.Anat Rec (Hoboken). 2008 Oct;291(10):1227-35. doi: 10.1002/ar.20736. Anat Rec (Hoboken). 2008. PMID: 18561194
-
Molecular control of lymphatic metastasis.Ann N Y Acad Sci. 2008;1131:225-34. doi: 10.1196/annals.1413.020. Ann N Y Acad Sci. 2008. PMID: 18519975 Review.
-
The role of the VEGF-C/VEGFR-3 axis in cancer progression.Br J Cancer. 2007 Feb 26;96(4):541-5. doi: 10.1038/sj.bjc.6603487. Epub 2006 Dec 12. Br J Cancer. 2007. PMID: 17164762 Free PMC article. Review.
Cited by
-
Proposal for a Histological Staging System of Mammary Carcinomas in Dogs and Cats. Part 2: Feline Mammary Carcinomas.Front Vet Sci. 2019 Nov 7;6:387. doi: 10.3389/fvets.2019.00387. eCollection 2019. Front Vet Sci. 2019. PMID: 31788484 Free PMC article.
-
Prognostic Insights in Feline Mammary Carcinomas: Clinicopathological Factors and the Proposal of a New Staging System.Animals (Basel). 2025 Mar 10;15(6):779. doi: 10.3390/ani15060779. Animals (Basel). 2025. PMID: 40150308 Free PMC article.
-
A Scoping Review on Tyrosine Kinase Inhibitors in Cats: Current Evidence and Future Directions.Animals (Basel). 2023 Sep 29;13(19):3059. doi: 10.3390/ani13193059. Animals (Basel). 2023. PMID: 37835664 Free PMC article.
-
Epithelial-to-Mesenchymal Transition and Phenotypic Marker Evaluation in Human, Canine, and Feline Mammary Gland Tumors.Animals (Basel). 2023 Feb 28;13(5):878. doi: 10.3390/ani13050878. Animals (Basel). 2023. PMID: 36899736 Free PMC article.
References
-
- Pepper MS. Lymphangiogenesis and tumor metastasis: myth or realty? Clinical Cancer Research. 2001;7:462–468. - PubMed
-
- Breitender-Geleff S, Soleiman A, Kowalski H, Horvat R, Amann G, Kriehuber E, Diem K, Weninger W, Tschachler E, Alitalo K, Kerjaschki D. Angiosarcomas express mixed endothelial phenotypes of blood and lymphatic capillaries: podoplanin as a specific marker for lymphatic endothelium. American Journal of Pathology. 1999;154:385–394. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

