Shared as well as distinct roles of EHD proteins revealed by biochemical and functional comparisons in mammalian cells and C. elegans
- PMID: 17233914
- PMCID: PMC1793994
- DOI: 10.1186/1471-2121-8-3
Shared as well as distinct roles of EHD proteins revealed by biochemical and functional comparisons in mammalian cells and C. elegans
Abstract
Background: The four highly homologous human EHD proteins (EHD1-4) form a distinct subfamily of the Eps15 homology domain-containing protein family and are thought to regulate endocytic recycling. Certain members of this family have been studied in different cellular contexts; however, a lack of concurrent analyses of all four proteins has impeded an appreciation of their redundant versus distinct functions.
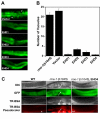
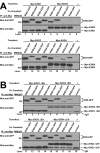
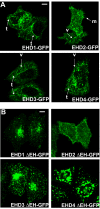
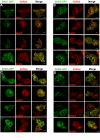
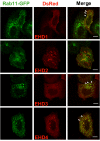
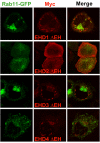
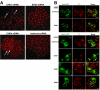
Results: Here, we analyzed the four EHD proteins both in mammalian cells and in a cross-species complementation assay using a C. elegans mutant lacking the EHD ortholog RME-1. We show that all human EHD proteins rescue the vacuolated intestinal phenotype of C. elegans rme-1 mutant, are simultaneously expressed in a panel of mammalian cell lines and tissues tested, and variably homo- and hetero-oligomerize and colocalize with each other and Rab11, a recycling endosome marker. Small interfering RNA (siRNA) knock-down of EHD1, 2 and 4, and expression of dominant-negative EH domain deletion mutants showed that loss of EHD1 and 3 (and to a lesser extent EHD4) but not EHD2 function retarded transferrin exit from the endocytic recycling compartment. EH domain deletion mutants of EHD1 and 3 but not 2 or 4, induced a striking perinuclear clustering of co-transfected Rab11. Knock-down analyses indicated that EHD1 and 2 regulate the exit of cargo from the recycling endosome while EHD4, similar to that reported for EHD3 (Naslavsky et al. (2006) Mol. Biol. Cell 17, 163), regulates transport from the early endosome to the recycling endosome.
Conclusion: Altogether, our studies suggest that concurrently expressed human EHD proteins perform shared as well as discrete functions in the endocytic recycling pathway and lay a foundation for future studies to identify and characterize the molecular pathways involved.
Figures




Similar articles
-
AMPH-1/Amphiphysin/Bin1 functions with RME-1/Ehd1 in endocytic recycling.Nat Cell Biol. 2009 Dec;11(12):1399-410. doi: 10.1038/ncb1986. Epub 2009 Nov 15. Nat Cell Biol. 2009. PMID: 19915558 Free PMC article.
-
Interactions between EHD proteins and Rab11-FIP2: a role for EHD3 in early endosomal transport.Mol Biol Cell. 2006 Jan;17(1):163-77. doi: 10.1091/mbc.e05-05-0466. Epub 2005 Oct 26. Mol Biol Cell. 2006. PMID: 16251358 Free PMC article.
-
EHD2 shuttles to the nucleus and represses transcription.Biochem J. 2012 Jun 15;444(3):383-94. doi: 10.1042/BJ20111268. Biochem J. 2012. PMID: 22448906
-
Mechanisms of EHD/RME-1 protein function in endocytic transport.Traffic. 2008 Dec;9(12):2043-52. doi: 10.1111/j.1600-0854.2008.00834.x. Epub 2008 Oct 14. Traffic. 2008. PMID: 18801062 Free PMC article. Review.
-
Molecular remodeling mechanisms of the neural somatodendritic compartment.Biochim Biophys Acta. 2012 Oct;1823(10):1720-30. doi: 10.1016/j.bbamcr.2012.06.006. Epub 2012 Jun 15. Biochim Biophys Acta. 2012. PMID: 22705351 Review.
Cited by
-
Caveolin-1 induces formation of membrane tubules that sense actomyosin tension and are inhibited by polymerase I and transcript release factor/cavin-1.Mol Biol Cell. 2010 Jul 1;21(13):2226-40. doi: 10.1091/mbc.e09-05-0417. Epub 2010 Apr 28. Mol Biol Cell. 2010. PMID: 20427576 Free PMC article.
-
Ferlin proteins in myoblast fusion and muscle growth.Curr Top Dev Biol. 2011;96:203-30. doi: 10.1016/B978-0-12-385940-2.00008-5. Curr Top Dev Biol. 2011. PMID: 21621072 Free PMC article. Review.
-
Endocytic recycling protein EHD1 regulates primary cilia morphogenesis and SHH signaling during neural tube development.Sci Rep. 2016 Feb 17;6:20727. doi: 10.1038/srep20727. Sci Rep. 2016. PMID: 26884322 Free PMC article.
-
EHD1 mediates vesicle trafficking required for normal muscle growth and transverse tubule development.Dev Biol. 2014 Mar 15;387(2):179-90. doi: 10.1016/j.ydbio.2014.01.004. Epub 2014 Jan 17. Dev Biol. 2014. PMID: 24440153 Free PMC article.
-
The function of EHD2 in endocytosis and defense signaling is affected by SUMO.Plant Mol Biol. 2014 Mar;84(4-5):509-18. doi: 10.1007/s11103-013-0148-7. Epub 2013 Oct 24. Plant Mol Biol. 2014. PMID: 24154852
References
-
- Wong WT, Kraus MH, Carlomagno F, Zelano A, Druck T, Croce CM, Huebner K, Di Fiore PP. The human eps15 gene, encoding a tyrosine kinase substrate, is conserved in evolution and maps to 1p31-p32. Oncogene. 1994;9:1591–1597. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 CA087986/CA/NCI NIH HHS/United States
- R01 CA096844/CA/NCI NIH HHS/United States
- CA96844/CA/NCI NIH HHS/United States
- CA81076/CA/NCI NIH HHS/United States
- R01 CA094143/CA/NCI NIH HHS/United States
- CA 87986/CA/NCI NIH HHS/United States
- CA94143/CA/NCI NIH HHS/United States
- CA 76118/CA/NCI NIH HHS/United States
- CA99163/CA/NCI NIH HHS/United States
- CA 99900/CA/NCI NIH HHS/United States
- R01 CA099900/CA/NCI NIH HHS/United States
- R01 CA081076/CA/NCI NIH HHS/United States
- T32 CA070085/CA/NCI NIH HHS/United States
- R01 CA099163/CA/NCI NIH HHS/United States
- T32 CA70085/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

