The sodium-coupled neutral amino acid transporter SNAT2 mediates an anion leak conductance that is differentially inhibited by transported substrates
- PMID: 17237199
- PMCID: PMC1864845
- DOI: 10.1529/biophysj.106.100776
The sodium-coupled neutral amino acid transporter SNAT2 mediates an anion leak conductance that is differentially inhibited by transported substrates
Abstract
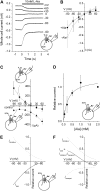
The sodium-coupled neutral amino acid transporter SNAT2 mediates cellular uptake of glutamine and other small, neutral amino acids. Here, we report the existence of a leak anion pathway associated with SNAT2. The leak anion conductance was increased by, but did not require the presence of, extracellular sodium. The transported substrates L-alanine, L-glutamine, and alpha-(methylamino)isobutyrate inhibited the anion leak conductance, each with different potency. A transporter with the mutation H-304A did not catalyze alanine transport but still catalyzed anion leak current, demonstrating that substrate transport is not required for anion current inhibition. Both the substrate and Na+ were able to bind to the SNAT2H-304A transporter normally. The selectivity sequence of the SNAT2H-304A anion conductance was SCN->>NO3->I->Br->Cl->Mes-. Anion flux mediated by the more hydrophobic anion SCN- was not saturable, whereas nitrate flux demonstrated saturation kinetics with an apparent Km of 29 mM. SNAT2, which belongs to the SLC38 family of transporters, has to be added to the growing number of secondary, Na+-coupled transporters catalyzing substrate-gated or leak anion conductances. Therefore, we can speculate that such anion-conducting pathways are general features of Na+-transporting systems.
Figures


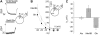
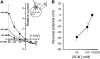
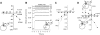


Similar articles
-
New inhibitors for the neutral amino acid transporter ASCT2 reveal its Na+-dependent anion leak.J Physiol. 2004 Jun 15;557(Pt 3):747-59. doi: 10.1113/jphysiol.2004.062521. Epub 2004 Apr 23. J Physiol. 2004. PMID: 15107471 Free PMC article.
-
Highly conserved asparagine 82 controls the interaction of Na+ with the sodium-coupled neutral amino acid transporter SNAT2.J Biol Chem. 2008 May 2;283(18):12284-92. doi: 10.1074/jbc.M706774200. Epub 2008 Mar 4. J Biol Chem. 2008. PMID: 18319257 Free PMC article.
-
Neutral amino acid transporter ASCT2 displays substrate-induced Na+ exchange and a substrate-gated anion conductance.Biochem J. 2000 Mar 15;346 Pt 3(Pt 3):705-10. Biochem J. 2000. PMID: 10698697 Free PMC article.
-
The role of the neutral amino acid transporter SNAT2 in cell volume regulation.Acta Physiol (Oxf). 2006 May-Jun;187(1-2):273-83. doi: 10.1111/j.1748-1716.2006.01552.x. Acta Physiol (Oxf). 2006. PMID: 16734764 Review.
-
Sodium-coupled neutral amino acid (System N/A) transporters of the SLC38 gene family.Pflugers Arch. 2004 Feb;447(5):784-95. doi: 10.1007/s00424-003-1117-9. Epub 2003 Jul 4. Pflugers Arch. 2004. PMID: 12845534 Review.
Cited by
-
Paternal obesity induces placental hypoxia and sex-specific impairments in placental vascularization and offspring metabolism†.Biol Reprod. 2022 Aug 9;107(2):574-589. doi: 10.1093/biolre/ioac066. Biol Reprod. 2022. PMID: 35377412 Free PMC article.
-
Functional expression of two system A glutamine transporter isoforms in rat auditory brainstem neurons.Neuroscience. 2009 Dec 15;164(3):998-1008. doi: 10.1016/j.neuroscience.2009.09.015. Epub 2009 Sep 12. Neuroscience. 2009. PMID: 19751803 Free PMC article.
-
Sodium-Dependent Neutral Amino Acid Transporter 2 Can Serve as a Tertiary Carrier for l-Type Amino Acid Transporter 1-Utilizing Prodrugs.Mol Pharm. 2023 Feb 6;20(2):1331-1346. doi: 10.1021/acs.molpharmaceut.2c00948. Epub 2023 Jan 23. Mol Pharm. 2023. PMID: 36688491 Free PMC article.
-
The C-terminal domain of the neutral amino acid transporter SNAT2 regulates transport activity through voltage-dependent processes.Biochem J. 2011 Mar 1;434(2):287-96. doi: 10.1042/BJ20100507. Biochem J. 2011. PMID: 21158741 Free PMC article.
-
Exploring Amino Acid Transporters as Therapeutic Targets for Cancer: An Examination of Inhibitor Structures, Selectivity Issues, and Discovery Approaches.Pharmaceutics. 2024 Jan 30;16(2):197. doi: 10.3390/pharmaceutics16020197. Pharmaceutics. 2024. PMID: 38399253 Free PMC article. Review.
References
-
- Johnson, L. W., and C. H. Smith. 1988. Neutral amino acid transport systems of microvillous membrane of human placenta. Am. J. Physiol. 254:C773–C780. - PubMed
-
- Sugawara, M., T. Nakanishi, Y. J. Fei, W. Huang, M. E. Ganapathy, F. H. Leibach, and V. Ganapathy. 2000. Cloning of an amino acid transporter with functional characteristics and tissue expression pattern identical to that of system A. J. Biol. Chem. 275:16473–16477. - PubMed
-
- Bode, B. P. 2001. Recent molecular advances in mammalian glutamine transport. J. Nutr. 131:2475S–2485S. - PubMed
-
- Yao, D., B. Mackenzie, H. Ming, H. Varoqui, H. Zhu, M. A. Hediger, and J. D. Erickson. 2000. A novel system A isoform mediating Na+/neutral amino acid cotransport. J. Biol. Chem. 275:22790–22797. - PubMed
-
- Shotwell, M. A., D. W. Jayme, M. S. Kilberg, and D. L. Oxender. 1981. Neutral amino acid transport systems in Chinese hamster ovary cells. J. Biol. Chem. 256:5422–5427. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

