Early disruptions of the blood-brain barrier may contribute to exacerbated neuronal damage and prolonged functional recovery following stroke in aged rats
- PMID: 17241702
- PMCID: PMC2683361
- DOI: 10.1016/j.neurobiolaging.2006.12.007
Early disruptions of the blood-brain barrier may contribute to exacerbated neuronal damage and prolonged functional recovery following stroke in aged rats
Abstract
We examined the effects of age on stroke progression and outcome in order to explore the association between blood-brain barrier (BBB) disruption, neuronal damage, and functional recovery. Using middle cerebral artery occlusion (MCAO), young (3 months) and aged (18 months) rats were assessed for BBB disruption at 20min post-MCAO, and 24h post-MCAO with tissue plasminogen activator induced reperfusion at 120min. Results showed that BBB disruptions in aged rats occurred early and increased nearly two-fold at both the 20min and 24h time points when compared to young animals. Neuronal damage in aged rats was increased two-fold as compared to young rats at 24h, while no neuronal damage was observed at 20min. Young and aged rats exhibited neurological deficits when compared to sham-controls out to 14 days following MCAO and reperfusion; however, aged rats exhibited more severe onset of deficits and prolonged recovery. Results indicate that aged rats suffer larger infarctions, reduced functional recovery and increased BBB disruption preceding observable neuronal injury.
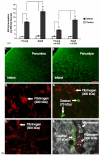
Figures




References
-
- AHA . Heart and stroke statistical update, 2000. American Heart Association; Dallas: 2001.
-
- Alkayed NJ, Harukuni I, Kimes AS, London ED, Traystman RJ, Hurn PD. Gender-linked brain injury in experimental stroke. Stroke. 1998;29:159–165. discussion 166. - PubMed
-
- Ariga T, Jarvis WD, Yu RK. Role of sphingolipid-mediated cell death in neurodegenerative diseases. J. Lipid Res. 1998;39:1–16. - PubMed
-
- Banks WA, Kastin AJ. Aging and the blood-brain barrier: changes in the carrier- mediated transport of peptides in rats. Neurosci. Lett. 1985;61:171–175. - PubMed
-
- Burns EM, Kruckeberg TW, Gaetano PK. Changes with age in cerebral capillary morphology. Neurobiol. Aging. 1981;2:283–291. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

