The function of guanylate cyclase 1 and guanylate cyclase 2 in rod and cone photoreceptors
- PMID: 17255100
- PMCID: PMC2043484
- DOI: 10.1074/jbc.M610369200
The function of guanylate cyclase 1 and guanylate cyclase 2 in rod and cone photoreceptors
Abstract
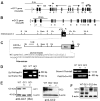
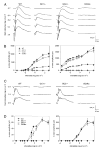
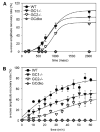
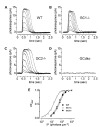
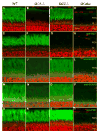
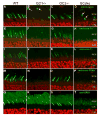
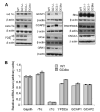
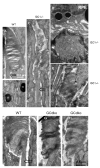
Retinal guanylate cyclases 1 and 2 (GC1 and GC2) are responsible for synthesis of cyclic GMP in rods and cones, but their individual contributions to phototransduction are unknown. We report here that the deletion of both GC1 and GC2 rendered rod and cone photoreceptors nonfunctional and unstable. In the rod outer segments of GC double knock-out mice, guanylate cyclase-activating proteins 1 and 2, and cyclic GMP phosphodiesterase were undetectable, although rhodopsin and transducin alpha-subunit were mostly unaffected. Outer segment membranes of GC1-/- and GC double knock-out cones were destabilized and devoid of cone transducin (alpha- and gamma-subunits), cone phosphodiesterase, and G protein-coupled receptor kinase 1, whereas cone pigments were present at reduced levels. Real time reverse transcription-PCR analyses demonstrated normal RNA transcript levels for the down-regulated proteins, indicating that down-regulation is posttranslational. We interpret these results to demonstrate an intrinsic requirement of GCs for stability and/or transport of a set of membrane-associated phototransduction proteins.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
- R01 EY006837/EY/NEI NIH HHS/United States
- R01 EY009339/EY/NEI NIH HHS/United States
- P30 EY011373/EY/NEI NIH HHS/United States
- EY08123/EY/NEI NIH HHS/United States
- R01 EY014596/EY/NEI NIH HHS/United States
- R37 EY006837/EY/NEI NIH HHS/United States
- EY06837/EY/NEI NIH HHS/United States
- F32 EY006837/EY/NEI NIH HHS/United States
- R01 EY008123/EY/NEI NIH HHS/United States
- P30 EY014800/EY/NEI NIH HHS/United States
- R01 DC006904/DC/NIDCD NIH HHS/United States
- EY09339/EY/NEI NIH HHS/United States
- P30 EY11373/EY/NEI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

