Uncrossed actions of feline corticospinal tract neurones on lumbar interneurones evoked via ipsilaterally descending pathways
- PMID: 17255170
- PMCID: PMC2075433
- DOI: 10.1113/jphysiol.2006.122739
Uncrossed actions of feline corticospinal tract neurones on lumbar interneurones evoked via ipsilaterally descending pathways
Abstract
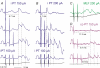
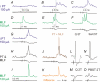
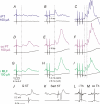
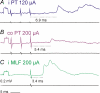
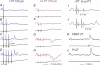
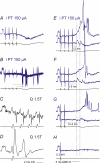
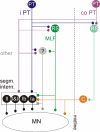
Effects of stimulation of ipsilateral pyramidal tract (PT) fibres were analysed in interneurones in midlumbar segments of the cat spinal cord in search of interneurones mediating disynaptic actions of uncrossed PT fibres on hindlimb motoneurones. The sample included 44 intermediate zone and ventral horn interneurones, most with monosynaptic input from group I and/or group II muscle afferents and likely to be premotor interneurones. Monosynaptic EPSPs evoked by stimulation of the ipsilateral PT were found in 12 of the 44 (27%) interneurones, while disynaptic or trisynaptic EPSPs were evoked in more than 75%. Both appeared at latencies that were either longer or within the same range as those of disynaptic EPSPs and IPSPs evoked by PT stimuli in motoneurones, making it unlikely that premotor interneurones in pathways from group I and/or II afferents relay the earliest actions of uncrossed PT fibres on motoneurones. These interneurones might nevertheless contribute to PT actions at longer latencies. Uncrossed PT actions on interneurones were to a great extent relayed via reticulospinal neurones with axons in the ipsilateral medial longitudinal fascicle (MLF), as indicated by occlusion and mutual facilitation of actions evoked by PT and MLF stimulation. However, PT actions were also relayed by other supraspinal or spinal neurones, as some remained after MLF lesions. Mutual facilitation and occlusion of actions evoked from the ipsilateral and contralateral PTs lead to the conclusion that the same midlumbar interneurones in pathways from group I or II muscle afferents may relay uncrossed and crossed PT actions.
Figures



Similar articles
-
Uncrossed actions of feline corticospinal tract neurones on hindlimb motoneurones evoked via ipsilaterally descending pathways.J Physiol. 2007 Apr 1;580(Pt 1):119-32. doi: 10.1113/jphysiol.2006.122721. Epub 2007 Jan 25. J Physiol. 2007. PMID: 17255171 Free PMC article.
-
Neuronal relays in double crossed pathways between feline motor cortex and ipsilateral hindlimb motoneurones.J Physiol. 2006 Sep 1;575(Pt 2):527-41. doi: 10.1113/jphysiol.2006.112425. Epub 2006 Jun 1. J Physiol. 2006. PMID: 16740611 Free PMC article.
-
Ipsilateral actions from the feline red nucleus on hindlimb motoneurones.J Physiol. 2008 Dec 15;586(24):5865-84. doi: 10.1113/jphysiol.2008.163998. Epub 2008 Oct 20. J Physiol. 2008. PMID: 18936076 Free PMC article.
-
Interactions between pathways controlling posture and gait at the level of spinal interneurones in the cat.Prog Brain Res. 1993;97:161-71. doi: 10.1016/s0079-6123(08)62274-8. Prog Brain Res. 1993. PMID: 8234742 Review.
-
Spinal interneuronal networks in the cat: elementary components.Brain Res Rev. 2008 Jan;57(1):46-55. doi: 10.1016/j.brainresrev.2007.06.022. Epub 2007 Aug 6. Brain Res Rev. 2008. PMID: 17884173 Free PMC article. Review.
Cited by
-
Bilateral postsynaptic actions of pyramidal tract and reticulospinal neurons on feline erector spinae motoneurons.J Neurosci. 2010 Jan 20;30(3):858-69. doi: 10.1523/JNEUROSCI.4859-09.2010. J Neurosci. 2010. PMID: 20089894 Free PMC article.
-
Premotor interneurones contributing to actions of feline pyramidal tract neurones on ipsilateral hindlimb motoneurones.J Physiol. 2008 Jan 15;586(2):557-74. doi: 10.1113/jphysiol.2007.145466. Epub 2007 Nov 15. J Physiol. 2008. PMID: 18006578 Free PMC article.
-
H-reflex down-conditioning greatly increases the number of identifiable GABAergic interneurons in rat ventral horn.Neurosci Lett. 2009 Mar 13;452(2):124-9. doi: 10.1016/j.neulet.2009.01.054. Epub 2009 Jan 24. Neurosci Lett. 2009. PMID: 19383426 Free PMC article.
-
Uncrossed actions of feline corticospinal tract neurones on hindlimb motoneurones evoked via ipsilaterally descending pathways.J Physiol. 2007 Apr 1;580(Pt 1):119-32. doi: 10.1113/jphysiol.2006.122721. Epub 2007 Jan 25. J Physiol. 2007. PMID: 17255171 Free PMC article.
-
Evidence for long-lasting subcortical facilitation by transcranial direct current stimulation in the cat.J Physiol. 2013 Jul 1;591(13):3381-99. doi: 10.1113/jphysiol.2012.244764. Epub 2013 Mar 18. J Physiol. 2013. PMID: 23507876 Free PMC article.
References
-
- Alstermark B, Isa T, Tantisira B. Pyramidal excitation in long propriospinal neurones in the cervical segments of the cat. Exp Brain Res. 1991;84:569–582. - PubMed
-
- Alstermark B, Lundberg A, Pinter M, Sasaki S. Subpopulations and functions of long C3–C5 propriospinal neurones. Brain Res. 1987;404:395–400. - PubMed
-
- Brookhart JM, Morris RE. Antidromic potential recordings from the bulbar pyramid of the cat. J Neurophysiol. 1948;11:387–398. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

