Phosphoinositide-dependent pathways in mouse sperm are regulated by egg ZP3 and drive the acrosome reaction
- PMID: 17258189
- PMCID: PMC1892180
- DOI: 10.1016/j.ydbio.2006.12.023
Phosphoinositide-dependent pathways in mouse sperm are regulated by egg ZP3 and drive the acrosome reaction
Abstract
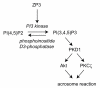
Sperm of many animals must complete an exocytotic event, the acrosome reaction, in order to fuse with eggs. In mammals, acrosome reactions are triggered during sperm contact with the egg extracellular matrix, or zona pellucida, by the matrix glycoprotein ZP3. Here, we show that ZP3 stimulates production of phosphatidylinositol-(3,4,5)-triphosphate in sperm membranes. Phosphatidylinositol-3-kinase antagonists that prevent acrosome reactions and fertilization in vitro, while generation of this phosphoinositide in the absence of ZP3 triggered acrosome reactions. Downstream effectors of phosphatidylinositol-(3,4,5)-triphosphate in sperm include the protein kinases, Akt and PKCzeta. These studies outline a signal transduction pathway that plays an essential role in the early events of mammalian fertilization.
Figures
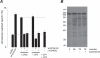

 ). PI3 kinase antagonists (100 nM wortmannin and 10 uM LY294002) had similar inhibitory effects on the ZP3/ZPse-evoked responses and data are pooled for presentation (
). PI3 kinase antagonists (100 nM wortmannin and 10 uM LY294002) had similar inhibitory effects on the ZP3/ZPse-evoked responses and data are pooled for presentation ( ). Similarly, control proteins (10 ug/ml ZP1 or ZP2; 20 ug/ml BSA, a1-acid glycoprotein) failed to evoke a phosphoinositide response and these results are pooled for presentation (
). Similarly, control proteins (10 ug/ml ZP1 or ZP2; 20 ug/ml BSA, a1-acid glycoprotein) failed to evoke a phosphoinositide response and these results are pooled for presentation ( ).
).
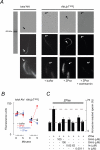
 , bpV(pic);
, bpV(pic);  , ZP3/ZPse;
, ZP3/ZPse;  , pbV(pic) + ZP3/ZPse. (B-D) Data represents the mean (± SD) of 5-7 separate experiments with >300 sperm assessed/experiment. Dashed lines in B-C represent minimum acrosome reaction levels in buffer-treated sperm and maximal signals in ZP3/ZPse-treated sperm, respectively. Dashed line in D represents the half-maximal acrosome reaction response. ZP3 and ZPse doses were 5 ug/ml and 20 ug/ml, respectively.
, pbV(pic) + ZP3/ZPse. (B-D) Data represents the mean (± SD) of 5-7 separate experiments with >300 sperm assessed/experiment. Dashed lines in B-C represent minimum acrosome reaction levels in buffer-treated sperm and maximal signals in ZP3/ZPse-treated sperm, respectively. Dashed line in D represents the half-maximal acrosome reaction response. ZP3 and ZPse doses were 5 ug/ml and 20 ug/ml, respectively.

Similar articles
-
Lewis X-containing neoglycoproteins mimic the intrinsic ability of zona pellucida glycoprotein ZP3 to induce the acrosome reaction in capacitated mouse sperm.Biol Reprod. 2004 Sep;71(3):778-89. doi: 10.1095/biolreprod.103.023820. Epub 2004 May 5. Biol Reprod. 2004. PMID: 15128591
-
Mammalian zona pellucida glycoproteins: structure and function during fertilization.Cell Tissue Res. 2012 Sep;349(3):665-78. doi: 10.1007/s00441-011-1319-y. Cell Tissue Res. 2012. PMID: 22298023 Review.
-
Regulating the acrosome reaction.Int J Dev Biol. 2008;52(5-6):503-10. doi: 10.1387/ijdb.082696hf. Int J Dev Biol. 2008. PMID: 18649263 Review.
-
Trp2 regulates entry of Ca2+ into mouse sperm triggered by egg ZP3.Nat Cell Biol. 2001 May;3(5):499-502. doi: 10.1038/35074570. Nat Cell Biol. 2001. PMID: 11331878
-
Ca(2+) entry through store-operated channels in mouse sperm is initiated by egg ZP3 and drives the acrosome reaction.Mol Biol Cell. 2000 May;11(5):1571-84. doi: 10.1091/mbc.11.5.1571. Mol Biol Cell. 2000. PMID: 10793136 Free PMC article.
Cited by
-
Reproductive toxicity of ritonavir in male: Insight into mouse sperm capacitation.Reprod Toxicol. 2022 Dec;114:1-6. doi: 10.1016/j.reprotox.2022.09.008. Epub 2022 Oct 2. Reprod Toxicol. 2022. PMID: 36198369 Free PMC article.
-
Toll-like Receptor 2 is Involved in Calcium Influx and Acrosome Reaction to Facilitate Sperm Penetration to Oocytes During in vitro Fertilization in Cattle.Front Cell Dev Biol. 2022 Feb 24;10:810961. doi: 10.3389/fcell.2022.810961. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 35281105 Free PMC article.
-
AMPK Function in Mammalian Spermatozoa.Int J Mol Sci. 2018 Oct 23;19(11):3293. doi: 10.3390/ijms19113293. Int J Mol Sci. 2018. PMID: 30360525 Free PMC article. Review.
-
Delineation of downstream signalling components during acrosome reaction mediated by heat solubilized human zona pellucida.Reprod Biol Endocrinol. 2010 Jan 23;8:7. doi: 10.1186/1477-7827-8-7. Reprod Biol Endocrinol. 2010. PMID: 20096131 Free PMC article.
-
Evidence that phosphatidylinositol 3-kinase is involved in sperm-induced tyrosine kinase signaling in Xenopus egg fertilization.BMC Dev Biol. 2009 Dec 17;9:68. doi: 10.1186/1471-213X-9-68. BMC Dev Biol. 2009. PMID: 20015408 Free PMC article.
References
-
- Amann RP, Hammerstedt RH. Detection of differences in fertility. J. Androl. 2002;23:317–325. - PubMed
-
- Aquila S, Sisci D, Gentile M, Middea E, Catalano S, Carpino A, Rago V, Ando S. Estrogen receptor (ER)alpha and ER beta are both expressed in human ejaculated spermatozoa: evidence of their direct interaction with phosphatidylinositol-3-OH kinase/Akt pathway. J. Clin. Endocrinol. Metab. 2004;89:1443–1451. - PubMed
-
- Bezzerides VJ, Ramsey IS, Kotecha S, Greka A, Clapham DE. Rapid vesicular translocation and insertion of TRP channels 5. Nat. Cell Biol. 2004;6:709–720. - PubMed
-
- Bleil JD, Wassarman PM. Sperm-egg interactions in the mouse: sequence of events and induction of the acrosome reaction by a zona pellucida glycoprotein. Dev. Biol. 1983;95:317–324. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

