Evaluation of the performance of the automated NucliSENS easyMAG and EasyQ systems versus the Roche AmpliPrep-AMPLICOR combination for high-throughput monitoring of human immunodeficiency virus load
- PMID: 17267632
- PMCID: PMC1865857
- DOI: 10.1128/JCM.01540-06
Evaluation of the performance of the automated NucliSENS easyMAG and EasyQ systems versus the Roche AmpliPrep-AMPLICOR combination for high-throughput monitoring of human immunodeficiency virus load
Abstract
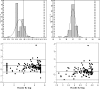
This study presents the data of an evaluation of the automated Nuclisens easyMAG and EasyQ systems versus the Roche AmpliPrep-AMPLICOR combination for testing of high-volume human immunodeficiency virus (HIV) load. This represents a follow-up of a previous study investigating the performance of the real-time Nuclisens assay using the semiautomated NucliSENS miniMAG extraction procedure. Three hundred eighteen patient samples were analyzed using both methods. The easyMAG-EasyQ HIV type 1 system has a higher sensitivity and broader dynamic range than the Cobas AmpliPrep-AMPLICOR system when the standard Roche assay is used alone, 25 to 3,000,000 IU/ml versus 400 to 750,000 HIV RNA copies/ml, respectively. There was significant correlation between the assays (0.93; P < 0.0001), with good accuracy (percent similarity mean mu = 96%), good precision (percent similarity standard deviation = 4.97%), and overall good agreement with a low percent similarity coefficient of variation of 5.17 to 6.11%. Bland-Altman analysis revealed that the AMPLICOR assay generated higher values than the EasyQ combination, with 95% of results within clinically acceptable limits. The throughput of samples was greatly improved using the easyMAG-EasyQ system, allowing 144 samples to be processed within 6 h. The potential for contamination has been dramatically reduced using the automated extraction system. Additional negative controls have been added to the kit to monitor for contamination based on the South African experience. This assay thus presents a real option for monitoring HIV load assays in high-volume testing environments.
Figures


Similar articles
-
Evaluation of NucliSens EasyQ HIV-1 assay for quantification of HIV-1 subtypes prevalent in South-east Asia.J Clin Virol. 2007 Jan;38(1):39-43. doi: 10.1016/j.jcv.2006.10.002. Epub 2006 Nov 15. J Clin Virol. 2007. PMID: 17110162
-
Evaluation of the NucliSens EasyQ assay in HIV-1-infected individuals in South Africa.J Virol Methods. 2005 Mar;124(1-2):105-10. doi: 10.1016/j.jviromet.2004.11.009. Epub 2004 Dec 15. J Virol Methods. 2005. PMID: 15664057
-
Evaluation of the Abbott m2000 RealTime human immunodeficiency virus type 1 (HIV-1) assay for HIV load monitoring in South Africa compared to the Roche Cobas AmpliPrep-Cobas Amplicor, Roche Cobas AmpliPrep-Cobas TaqMan HIV-1, and BioMerieux NucliSENS EasyQ HIV-1 assays.J Clin Microbiol. 2009 Jul;47(7):2209-17. doi: 10.1128/JCM.01761-08. Epub 2009 May 6. J Clin Microbiol. 2009. PMID: 19420172 Free PMC article.
-
Comparison between the automated Roche Cobas AmpliPrep/Cobas TaqMan HIV-1 test version 2.0 assay and its version 1 and Nuclisens HIV-1 EasyQ version 2.0 assays when measuring diverse HIV-1 genotypes in China.J Clin Virol. 2012 Jan;53(1):33-7. doi: 10.1016/j.jcv.2011.10.001. Epub 2011 Nov 2. J Clin Virol. 2012. PMID: 22051503
-
Multicenter evaluation of the NucliSens EasyQ HIV-1 v1.1 assay for the quantitative detection of HIV-1 RNA in plasma.J Virol Methods. 2005 Jul;127(1):54-9. doi: 10.1016/j.jviromet.2005.03.013. Epub 2005 Apr 25. J Virol Methods. 2005. PMID: 15893566
Cited by
-
Systematic review of the performance of HIV viral load technologies on plasma samples.PLoS One. 2014 Feb 18;9(2):e85869. doi: 10.1371/journal.pone.0085869. eCollection 2014. PLoS One. 2014. PMID: 24558359 Free PMC article.
-
Does Size Matter? Comparison of Extraction Yields for Different-Sized DNA Fragments by Seven Different Routine and Four New Circulating Cell-Free Extraction Methods.J Clin Microbiol. 2018 Nov 27;56(12):e01061-18. doi: 10.1128/JCM.01061-18. Print 2018 Dec. J Clin Microbiol. 2018. PMID: 30282788 Free PMC article.
-
Evaluation of the Cavidi ExaVir Load assay (version 3) for plasma human immunodeficiency virus type 1 load monitoring.J Clin Microbiol. 2009 Sep;47(9):3011-3. doi: 10.1128/JCM.00805-09. Epub 2009 Jul 15. J Clin Microbiol. 2009. PMID: 19605583 Free PMC article.
-
Evaluation of DNA Extraction Methods for Reliable Quantification of Acinetobacter baumannii, Klebsiella pneumoniae, and Pseudomonas aeruginosa.Biosensors (Basel). 2023 Apr 6;13(4):463. doi: 10.3390/bios13040463. Biosensors (Basel). 2023. PMID: 37185538 Free PMC article.
-
Clinical evaluation of NucliSENS magnetic extraction and NucliSENS analyte-specific reagents for real-time detection of human metapneumovirus in pediatric respiratory specimens.J Clin Microbiol. 2008 Apr;46(4):1274-80. doi: 10.1128/JCM.01567-07. Epub 2008 Feb 13. J Clin Microbiol. 2008. PMID: 18272716 Free PMC article.
References
-
- Bland, J. M., and D. G. Altman. 1986. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet i:307-310. - PubMed
-
- de Mendoza, C., M. Koppelman, B. Montes, V. Ferre, V. Soriano, H. Cuypers, M. Segondy, and T. Oosterlaken. 2005. Multicenter evaluation of the NucliSens EasyQ HIV-1 v1.1 assay for the quantitative detection of HIV-1 RNA in plasma. J. Virol. Methods 127:54-59. - PubMed
-
- National Department of Health. 2004. National antiretroviral treatment guidelines. National Department of Health, Johannesburg, South Africa. http://www.doh.gov.za/docs/factsheets/guidelines/artguide04-f.html.
-
- Scott, L. E., J. S. Galpin, and D. K. Glencross. 2003. Multiple method comparison: statistical model using percentage similarity. Cytometry B Clin. Cytom. 54:46-53. - PubMed
-
- Stevens, W., T. Wiggill, P. Horsfield, L. Coetzee, and L. E. Scott. 2005. Evaluation of the NucliSens EasyQ assay in HIV-1-infected individuals in South Africa. J. Virol. Methods 124:105-110. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

