Evaluation of an LC8-binding peptide for the attachment of artificial cargo to dynein
- PMID: 17274669
- PMCID: PMC2569131
- DOI: 10.1021/mp060086o
Evaluation of an LC8-binding peptide for the attachment of artificial cargo to dynein
Abstract
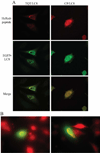
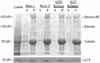
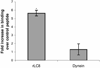
The limited cytoplasmic mobility of nonviral gene carriers is likely to contribute to their low transfection efficiency. This limitation could be overcome by mimicking the viral strategy of recruiting the dynein motor complex for efficient transport toward the host cell nucleus. A promising approach for attaching artificial cargo to dynein is through an adaptor peptide that binds the 8 kDa light chain (LC8) found in the cargo-binding region of the dynein complex. Several viral proteins that bind LC8 have in common an LC8-binding motif defined by (K/R)XTQT. Short peptides containing this motif have also been shown to bind recombinant LC8 in vitro. However, since the majority of intracellular LC8 exists outside of the dynein complex, it remains unclear whether peptides displaying this LC8-binding motif can access and bind to dynein-associated LC8. In this study, we employed biochemical analysis to investigate the feasibility of attaching artificial cargo to the dynein motor complex using a peptide displaying the well-characterized LC8-binding motif. We report that free intracellular LC8 bound specifically to an LC8-binding (TQT) peptide and not to a control peptide with a mutated LC8-binding motif. However, a similar binding interaction between the TQT peptide and intracellular dynein was not detected. To determine whether dynein binding of the TQT peptide was prevented by competition with free intracellular LC8 or due to the inability of the peptide to access its LC8 binding site in the dynein complex, the TQT peptide was evaluated for its ability to bind either purified LC8 or purified dynein. Our results demonstrate that, while the TQT peptide readily binds free LC8, it cannot bind to dynein-associated LC8. The results emphasize the need to identify functional dynein-binding peptides and highlight the importance of designing peptides that bind to the intact dynein motor complex.
Figures


References
-
- Lukacs GL, Haggie P, Seksek O, Lechardeur D, Freedman N, Verkman AS. Sizedependent DNA mobility in cytoplasm and nucleus. J Biol Chem. 2000;275:1625–9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

