Remedial strategies in structural proteomics: expression, purification, and crystallization of the Vav1/Rac1 complex
- PMID: 17275330
- PMCID: PMC1892187
- DOI: 10.1016/j.pep.2006.10.027
Remedial strategies in structural proteomics: expression, purification, and crystallization of the Vav1/Rac1 complex
Abstract
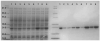
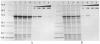
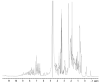
The signal transduction pathway involving the Vav1 guanine nucleotide exchange factor (GEF) and the Rac1 GTPase plays several key roles in the immune response mediated by the T cell receptor. Vav1 is also a unique member of the GEF family in that it contains a cysteine-rich domain (CRD) that is critical for Rac1 binding and maximal guanine nucleotide exchange activity, and thus may provide a unique protein-protein interface compared to other GEF/GTPase pairs. Here, we have applied a number of remedial structural proteomics strategies, such as construct and expression optimization, surface mutagenesis, limited proteolysis, and protein formulation to successfully express, purify, and crystallize the Vav1-DH-PH-CRD/Rac1 complex in an active conformation. We have also systematically characterized various Vav1 domains in a GEF assay and Rac1 in vitro binding experiments. In the context of Vav1-DH-PH-CRD, the zinc finger motif of the CRD is required for the expression of stable Vav1, as well as for activity in both a GEF assay and in vitro formation of a Vav1/Rac1 complex suitable for biophysical and structural characterization. Our data also indicate that the isolated CRD maintains a low level of specific binding to Rac1, appears to be folded based on 1D NMR analysis and coordinates two zinc ions based on ICP-MS analysis. The protein reagents generated here are essential tools for the determination of a three dimensional Vav1/Rac1 complex crystal structure and possibly for the identification of inhibitors of the Vav1/Rac1 protein-protein interaction with potential to inhibit lymphocyte activation.
Figures


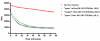
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

