Neuropeptide Y acts at Y1 receptors in the rostral ventral medulla to inhibit neuropathic pain
- PMID: 17276005
- PMCID: PMC2077302
- DOI: 10.1016/j.pain.2006.12.018
Neuropeptide Y acts at Y1 receptors in the rostral ventral medulla to inhibit neuropathic pain
Abstract
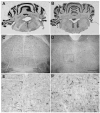
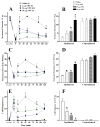
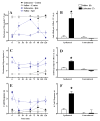
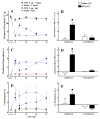
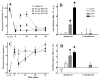
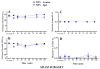
Brain microinjection studies in the rat using local anesthetics suggest that the rostral ventral medulla (RVM) contributes to the facilitation of neuropathic pain. However, these studies were restricted to a single model of neuropathic pain (the spinal nerve ligation model) and to just two stimulus modalities (non-noxious tactile stimulus and heat). Also, few neurotransmitter systems have been shown to modulate descending facilitation. After either partial sciatic nerve ligation (PSNL) or spared nerve injury (SNI), we found that unilateral or bilateral microinjection of lidocaine into the RVM reduced not only mechanical allodynia (decreased threshold to von Frey hairs and/or an automated device) and mechanical hyperalgesia (increased paw lifting in response to a noxious pin), but also cold hypersensitivity (increased lifting in response to the hindpaw application of a drop of acetone). Application of a drop of water did not elicit paw withdrawal, indicating that the acetone test is indeed a measure of cold hypersensitivity. We found significant neuropeptide Y Y1-like immunoreactivity within, and lateral to, the midline RVM. Intra-RVM injection of neuropeptide Y (NPY) dose-dependently inhibited the mechanical and cold hypersensitivity associated with PSNL or SNI, an effect that could be blocked by the Y1 receptor antagonist BIBO 3304. We conclude that medullary facilitation spans multiple behavioral signs of allodynia and hyperalgesia in multiple models of neuropathic pain. Furthermore, NPY inhibits behavioral signs of neuropathic pain, possibly by acting at Y1 receptors in the RVM.
Figures


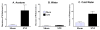
Similar articles
-
Neuropeptide Y in the rostral ventromedial medulla reverses inflammatory and nerve injury hyperalgesia in rats via non-selective excitation of local neurons.Neuroscience. 2014 Jun 20;271:149-59. doi: 10.1016/j.neuroscience.2014.04.035. Epub 2014 Apr 30. Neuroscience. 2014. PMID: 24792711 Free PMC article.
-
Intrathecal neuropeptide Y reduces behavioral and molecular markers of inflammatory or neuropathic pain.Pain. 2008 Jul 15;137(2):352-365. doi: 10.1016/j.pain.2007.09.016. Epub 2007 Oct 31. Pain. 2008. PMID: 17976913 Free PMC article.
-
Selective mediation of nerve injury-induced tactile hypersensitivity by neuropeptide Y.J Neurosci. 2002 Nov 15;22(22):9858-67. doi: 10.1523/JNEUROSCI.22-22-09858.2002. J Neurosci. 2002. PMID: 12427842 Free PMC article.
-
Spinal activation of the NPY Y1 receptor reduces mechanical and cold allodynia in rats with chronic constriction injury.Peptides. 2017 Jun;92:38-45. doi: 10.1016/j.peptides.2017.04.005. Epub 2017 Apr 29. Peptides. 2017. PMID: 28465077
-
Inflammation enhances Y1 receptor signaling, neuropeptide Y-mediated inhibition of hyperalgesia, and substance P release from primary afferent neurons.Neuroscience. 2014 Jan 3;256:178-94. doi: 10.1016/j.neuroscience.2013.10.054. Epub 2013 Oct 31. Neuroscience. 2014. PMID: 24184981 Free PMC article.
Cited by
-
The left central nucleus of the amygdala contributes to mechanical allodynia and hyperalgesia following right-sided peripheral nerve injury.Neurosci Lett. 2018 Sep 25;684:187-192. doi: 10.1016/j.neulet.2018.08.013. Epub 2018 Aug 13. Neurosci Lett. 2018. PMID: 30114475 Free PMC article.
-
Neuropeptide Y and Pain: Insights from Brain Research.ACS Pharmacol Transl Sci. 2024 Nov 2;7(12):3718-3728. doi: 10.1021/acsptsci.4c00333. eCollection 2024 Dec 13. ACS Pharmacol Transl Sci. 2024. PMID: 39698268 Review.
-
Fasting diets: what are the impacts on eating behaviors, sleep, mood, and well-being?Front Nutr. 2024 Jan 9;10:1256101. doi: 10.3389/fnut.2023.1256101. eCollection 2023. Front Nutr. 2024. PMID: 38264193 Free PMC article. Review.
-
Pioglitazone rapidly reduces neuropathic pain through astrocyte and nongenomic PPARγ mechanisms.Pain. 2015 Mar;156(3):469-482. doi: 10.1097/01.j.pain.0000460333.79127.be. Pain. 2015. PMID: 25599238 Free PMC article.
-
Modality selective roles of pro-nociceptive spinal 5-HT2A and 5-HT3 receptors in normal and neuropathic states.Neuropharmacology. 2018 Dec;143:29-37. doi: 10.1016/j.neuropharm.2018.09.028. Epub 2018 Sep 18. Neuropharmacology. 2018. PMID: 30240783 Free PMC article.
References
-
- Chaplan SR, Bach FW, Pogrel JW, Chung JM, Yaksh TL. Quantitative assessment of tactile allodynia in the rat paw. Journal of Neuroscience Methods. 1994;53:55–63. - PubMed
-
- Chen Q, King T, Vanderah TW, Ossipov MH, Malan TP, Jr, Lai J, Porreca F. Differential blockade of nerve injury-induced thermal and tactile hypersensitivity by systemically administered brain-penetrating and peripherally restricted local anesthetics. J Pain. 2004;5(5):281–289. - PubMed
-
- Decosterd I, Allchorne A, Woolf CJ. Differential analgesic sensitivity of two distinct neuropathic pain models. Anesth Analg. 2004;99(2):457–463. table of contents. - PubMed
-
- Decosterd I, Woolf CJ. Spared nerve injury: an animal model of persistent peripheral neuropathic pain. Pain. 2000;87(2):149–158. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

