Environmental lead exposure during early life alters granule cell neurogenesis and morphology in the hippocampus of young adult rats
- PMID: 17276012
- PMCID: PMC1892316
- DOI: 10.1016/j.neuroscience.2006.12.040
Environmental lead exposure during early life alters granule cell neurogenesis and morphology in the hippocampus of young adult rats
Abstract
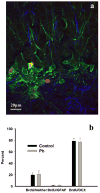
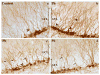
Exposure to environmentally relevant levels of lead (Pb(2+)) during early life produces deficits in hippocampal synaptic plasticity in the form of long-term potentiation (LTP) and spatial learning in young adult rats [Nihei MK, Desmond NL, McGlothan JL, Kuhlmann AC, Guilarte TR (2000) N-methyl-D-aspartate receptor subunit changes are associated with lead-induced deficits of long-term potentiation and spatial learning. Neuroscience 99:233-242; Guilarte TR, Toscano CD, McGlothan JL, Weaver SA (2003) Environmental enrichment reverses cognitive and molecular deficits induced by developmental lead exposure. Ann Neurol 53:50-56]. Other evidence suggests that the performance of rats in the Morris water maze spatial learning tasks is associated with the level of granule cell neurogenesis in the dentate gyrus (DG) [Drapeau E, Mayo W, Aurousseau C, Le Moal M, Piazza P-V, Abrous DN (2003) Spatial memory performance of aged rats in the water maze predicts level of hippocampal neurogenesis. Proc Natl Acad Sci U S A 100:14385-14390]. In this study, we examined whether continuous exposure to environmentally relevant levels of Pb(2+) during early life altered granule cell neurogenesis and morphology in the rat hippocampus. Control and Pb(2+)-exposed rats received bromodeoxyuridine (BrdU) injections (100 mg/kg; i.p.) for five consecutive days starting at postnatal day 45 and were killed either 1 day or 4 weeks after the last injection. The total number of newborn cells in the DG of Pb(2+)-exposed rats was significantly decreased (13%; P<0.001) 1 day after BrdU injections relative to controls. Further, the survival of newborn cells in Pb(2+)-exposed rats was significantly decreased by 22.7% (P<0.001) relative to control animals. Co-localization of BrdU with neuronal or astrocytic markers did not reveal a significant effect of Pb(2+) exposure on cellular fate. In Pb(2+)-exposed rats, immature granule cells immunolabeled with doublecortin (DCX) displayed aberrant dendritic morphology. That is, the overall length-density of the DCX-positive apical dendrites in the outer portion of the DG molecular layer was significantly reduced up to 36% in the suprapyramidal blade only. We also found that the area of Timm's-positive staining representative of the mossy fibers terminal fields in the CA3 stratum oriens (SO) was reduced by 26% in Pb(2+)-exposed rats. These findings demonstrate that exposure to environmentally relevant levels of Pb(2+) during early life alters granule cell neurogenesis and morphology in the rat hippocampus. They provide a cellular and morphological basis for the deficits in synaptic plasticity and spatial learning documented in Pb(2+)-exposed animals.
Figures
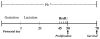




Similar articles
-
Chronic developmental lead exposure reduces neurogenesis in adult rat hippocampus but does not impair spatial learning.Toxicol Sci. 2005 Aug;86(2):365-74. doi: 10.1093/toxsci/kfi156. Epub 2005 Mar 23. Toxicol Sci. 2005. PMID: 15788721
-
Hippocampal expression of N-methyl-D-aspartate receptor (NMDAR1) subunit splice variant mRNA is altered by developmental exposure to Pb(2+).Brain Res Mol Brain Res. 2000 Mar 29;76(2):299-305. doi: 10.1016/s0169-328x(00)00010-3. Brain Res Mol Brain Res. 2000. PMID: 10762705
-
Ketamine Affects the Neurogenesis of the Hippocampal Dentate Gyrus in 7-Day-Old Rats.Neurotox Res. 2016 Aug;30(2):185-98. doi: 10.1007/s12640-016-9615-7. Epub 2016 Mar 10. Neurotox Res. 2016. PMID: 26966008
-
Glutamatergic components underlying lead-induced impairments in hippocampal synaptic plasticity.Neurotoxicology. 2000 Dec;21(6):1057-68. Neurotoxicology. 2000. PMID: 11233752 Review.
-
New and evolving concepts in the neurotoxicology of lead.Toxicol Appl Pharmacol. 2007 Nov 15;225(1):1-27. doi: 10.1016/j.taap.2007.08.001. Epub 2007 Aug 16. Toxicol Appl Pharmacol. 2007. PMID: 17904601 Review.
Cited by
-
Prenatal lead exposure and schizophrenia: further evidence and more neurobiological connections.Environ Health Perspect. 2009 May;117(5):A190-1. doi: 10.1289/ehp.0800484. Environ Health Perspect. 2009. PMID: 19478978 Free PMC article. No abstract available.
-
From the Cover: 7,8-Dihydroxyflavone Rescues Lead-Induced Impairment of Vesicular Release: A Novel Therapeutic Approach for Lead Intoxicated Children.Toxicol Sci. 2018 Jan 1;161(1):186-195. doi: 10.1093/toxsci/kfx210. Toxicol Sci. 2018. PMID: 29029315 Free PMC article.
-
Ginsenoside Rd maintains adult neural stem cell proliferation during lead-impaired neurogenesis.Neurol Sci. 2013 Jul;34(7):1181-8. doi: 10.1007/s10072-012-1215-6. Epub 2012 Oct 17. Neurol Sci. 2013. PMID: 23073826
-
Cerebral Vascular Toxicity after Developmental Exposure to Arsenic (As) and Lead (Pb) Mixtures.Toxics. 2024 Aug 24;12(9):624. doi: 10.3390/toxics12090624. Toxics. 2024. PMID: 39330552 Free PMC article.
-
Lead exposure and fear-potentiated startle in the VA Normative Aging Study: a pilot study of a novel physiological approach to investigating neurotoxicant effects.Neurotoxicol Teratol. 2013 Jul-Aug;38:21-8. doi: 10.1016/j.ntt.2013.04.003. Epub 2013 Apr 17. Neurotoxicol Teratol. 2013. PMID: 23603705 Free PMC article.
References
-
- Abrous DN, Koehl M, Le Moal M. Adult neurogenesis: from precursors to network and physiology. Physiol Rev. 2005;85:523–569. - PubMed
-
- Alkondon M, Costa AC, Radhakrishnan V, Aronstam RS, Albuquerque EX. Selective blockade of NMDA-activated channel currents may be implicated in learning deficits caused by lead. FEBS Lett. 1990;261:124–130. - PubMed
-
- Ambrogini P, Lattanzi D, Ciuffoli S, Agostini D, Bertini L, Stocchi V, Santi S, Cuppini R. Morpho-functional characterization of neuronal cells at different stages of maturation in granule cell layer of adult rat dentate gyrus. Brain Res. 2004;1017:21–31. - PubMed
-
- Arvidsson A, Kokaia Z, Lindvall O. N-methyl-D-aspartate receptor-mediated increase of neurogenesis in adult dentate gyrus following stroke. Eur J Neurosci. 2001;14:10–18. - PubMed
-
- Bannigan JG. Autoradiographic analysis of effects of 5-bromodeoxyuridine on neurogenesis in the chick embryo spinal cord. Brain Res. 1987;433:161–170. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

